Bromocresol Green Indicator
Bromocresol green (BCG) is a dye that is used in chemistry as a pH indicator in acid-base titration and in thin-layer chromatography staining solutions to visualize acidic compounds. The aqueous indicator solution of bromocresol green is yellow below pH 3.8 and blue above pH 5.4. It is also important in clinical diagnosis for measuring serum albumin concentration within mammalian blood samples and DNA agarose gel electrophoresis.
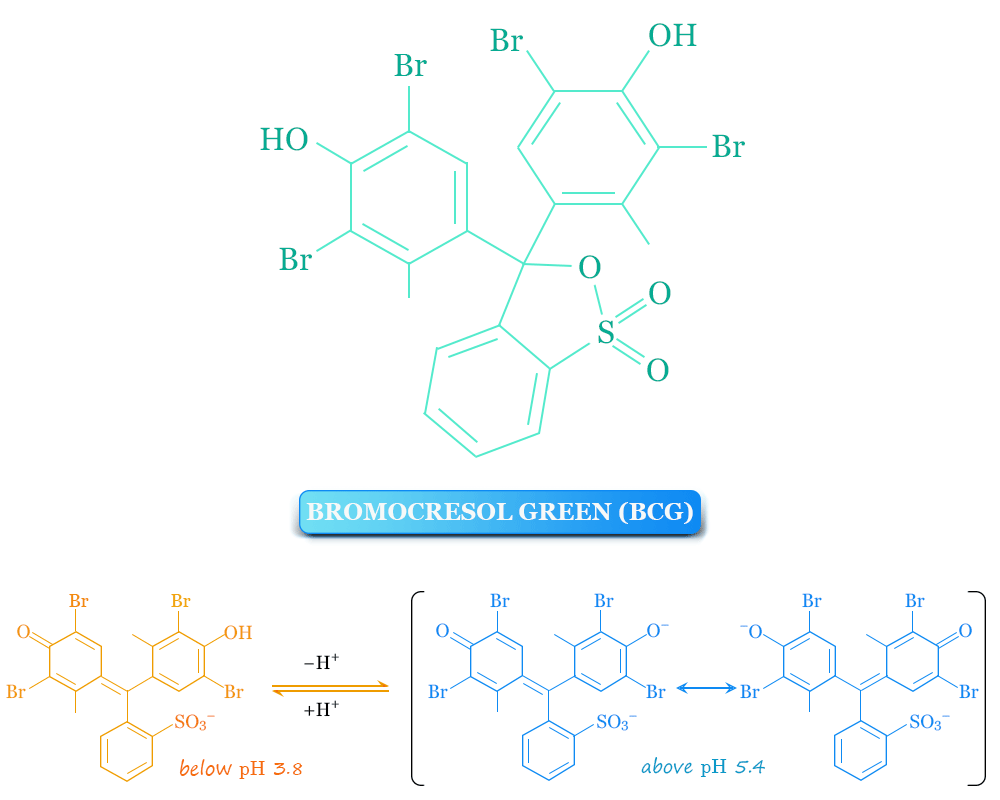
The chemical formula of bromocresol green is C21H14Br4O5S. The aqueous solution of bromocresol green indicator is yellow below pH 3.8 and blue above pH 5.4. The green colour of the indicator shows between pH 3.8 and 5.4.
Properties
Bromocresol green indicator is an odorless, light yellow-purple solid available in the market as a free acid form, monosodium salt, and dilute solution.
BCG is a pH-sensitive dye belonging to the family of triarylmethane dyes. It is a beige to brown solid powder or crystals that show a yellow color below pH of 3.8 and a blue color above pH of 5.4. Therefore, it is used in chemistry as a pH indicator.
Some common properties of bromocresol green are listed below in the table:
| Bromocresol Green | |
| IUPAC name | 3,3-Bis(3,5-dibromo-4-hydroxy-2-methylphenyl)-2,1λ6-benzoxathiole-1,1(3H)-dione |
| Molecular Formula | C21H14Br4O5S |
| Molecular Weight | 698.0 g/mol |
| CAS No | 76-60-8 |
| Appearance | Beige to brown solid powder or crystals |
| Solubility | Sparingly soluble in water, highly soluble in ethanol and diethyl ether, and soluble in benzene |
| Melting Point | 225 °C |
| Acidity (pKa) | 4.90 |
| UV-vis (λmax) | 423 nm |
BCG can be ionized in an aqueous solution to give the monoanionic yellow form which further deprotonates at a higher pH range to give the dianionic blue form of the indicator. The dianionic blue form of the indicator is stabilized by resonance.
The acidic and basic forms of BCG dye have an isosbestic point in their UV-visible spectrum, around 515 nm. Therefore, it indicates that the two forms of BCG can be interconverted directly without forming any other substance.
Preparation
For the preparation of bromocresol green indicator solution, we need soluble solvents such as deionized water or ethyl alcohol (ethanol). Generally, the indicator solution is made by dissolving bromocresol green powder in ethanol. However, we can prepare this indicator solution in two ways.
- 0.1% alcohol solution: Dissolve 0.1 g of indicator in 75 mL of ethyl alcohol and dilute the solution with ethyl alcohol to make 100 ml. We can also use diethyl ether or benzene in place of ethyl alcohol.
- 0.04% aqueous solution: Dissolve 0.04 g of bromocresol green in 50 mL of deionized water and dilute the solution with water to make 100 ml.
Uses of Bromocresol Green pH Indicator
Bromocresol green (BCG) is a triphenylmethane dye that has various applications in chemistry, analytical chemistry, biochemistry, and medicine.
- BCG is a pH-sensitive dye that is used in chemistry as a pH indicator. It is also used as a pH indicator in applications such as growth mediums for microorganisms and titrations.
- In thin-layer chromatography (TLC), BCG is put for visualizing molecules that contain acidic compounds with pKa values lower than 5.
- In biochemical analysis, it is an active tracking dye for DNA Agarose gel electrophoresis.
- BCG is used in clinical diagnosis for the determination of the concentration of serum albumin present in mammalian blood samples taken from patients suspected of having kidney failure or liver disease.
- In medicinal chemistry, it acts as an inhibitor of the protein that transports prostaglandin E2.
- Additionally, it can be used to detect ammonia and utilization in sol-gel matrices. Therefore, ammonia vapour is often used for improving the sensitivity of visualization of organic acids where bromocresol green or bromophenol blue pH indicators have been used initially.
Safety Information
Contact BCG powder or indicator may cause respiratory tract and skin irritation. It also causes serious eye irritation. Therefore, contact with human skin, mucous membranes, and eyes should be harmful and avoided.
Frequently Asked Questions (FAQ)
How does BCG work with albumin?
Bromcresol Green (BCG) is a known dye for its ability to bind many types of organic compounds. Albumin in the presence of BCG shows a slightly acidic pH. Therefore, the BCG indicator changes colour from yellow-green to green-blue.
When albumin binds with BCG, it causes a change in the absorbance maximum of the BCG indicator. Such type of change can be measured spectrophotometrically and used to determine albumin concentration.
Is bromocresol green a weak acid?
The acid-base indicator bromocresol green is a weak acid with a pKa of 4.90. Therefore, it is more acidic than phenol and similar to acetic acid. The yellow acidic and blue basic forms of the BCG indicator are present in equal concentrations in a solution when the pH is 4.38.
What pH does the bromocresol green indicator change color?
BCG (chemical name 3′, 3”, 5′, 5”-tetrabromo-m-cresolsulfonphthalein) is a sulfonphthalein dye that has a transition range of pH 3.8 to 5.4.
In the acidic form (below pH 3.8), the bromocresol green indicator appears yellow and in the basic form (above pH 5.4), it is blue. The green colour of the BCG indicator shows between pH 3.8 and 5.4.