Periodic Table of Elements
Periodic table elements arrangement in chemistry in order of increasing atomic number or the full number of protons in the atomic nucleus is named as modern law of the periodic table. According to the modern law or long form of the periodic table, the list of chemical elements along groups or periods is organized on the basis of electronic configuration and classified into four categories s, p, d, and f-block elements. The modern law for the periodic table of elements suggests that the physical and chemical properties of elements are the periodic functions of the atomic number. The modern law of the periodic table comes due to the breakdown of Mendeleev’s (1861) scientific classification based on atomic weight or masses of the chemical elements. The names, symbols, atomic numbers, and electronic configuration of 118 periodic table elements are given below in the picture:
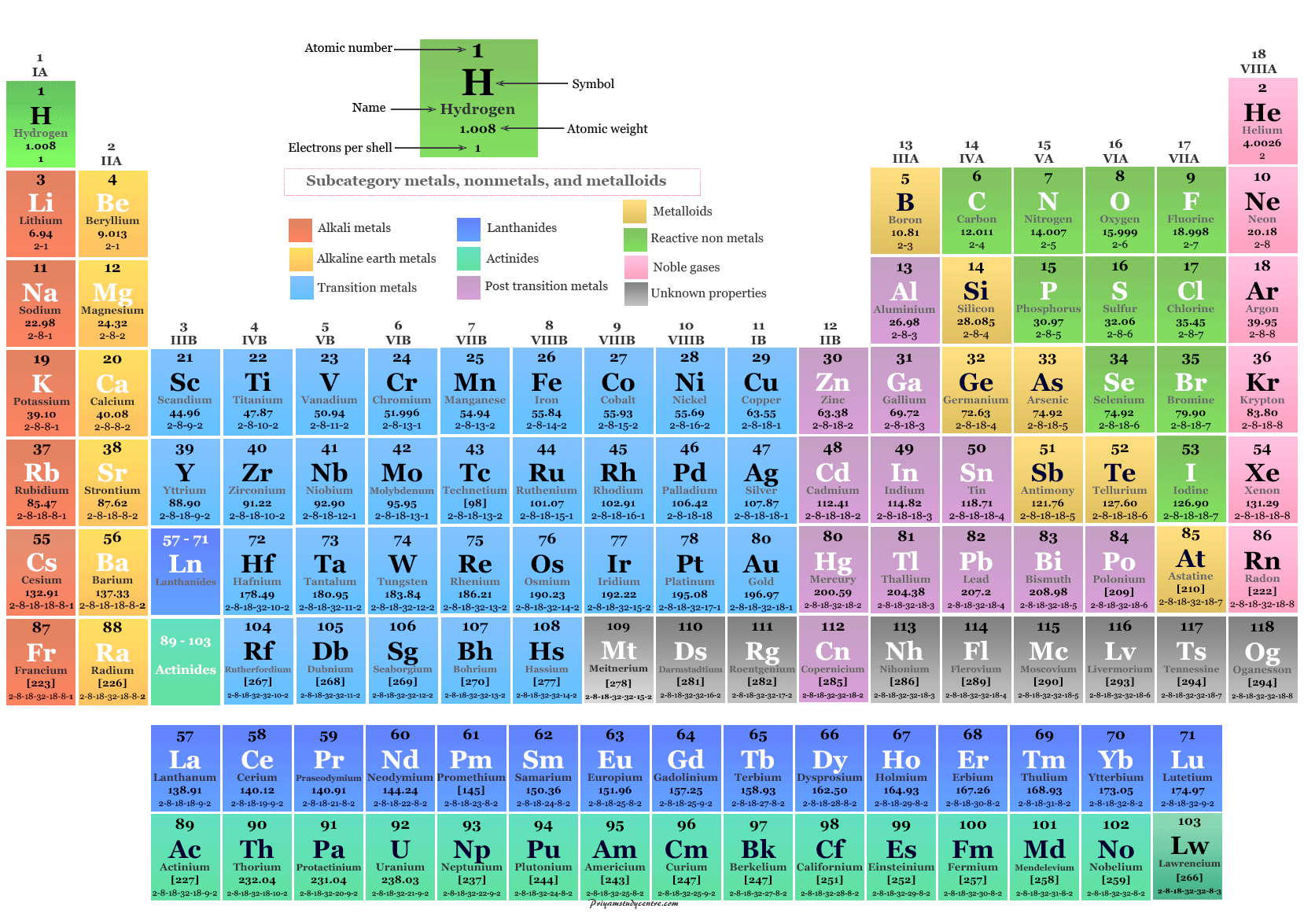
Modern Periodic Table
The modern law for the periodic table of elements comes to remove the defects of Mendeleev Mendeleev’s scientific classification. In the modern periodic table chart, the elements are represented by two parts, vertical columns (group) and horizontal rows (period) in chemistry or chemical science
The initial discovery was explained by Dmitri Ivanovich Mendeleev in 1861 and Mosely in 1911. However, the Bohr model or scheme suggested the scientific development of groups and periods of the periodic table elements.
Periods in Periodic Table of Elements
The periods on the periodic table are the horizontal rows used to arrange the chemical elements. The long form or modern form of the periodic table contains seven (7) periods for the accommodation of elements.
First Period
The principal quantum number (n) = 1 indicates that there is only one main energy level for the elements in this period. Therefore, this period has two chemical elements beginning with hydrogen and ending with inert gas helium.
Second Period
There are two sub-shells (2s and 2p) for the elements in this period. Therefore, this period has (2×1 + 3×2) = 8 chemical elements beginning with the alkali metal lithium and ending with the inert gas neon.
Third Period
There are two sub-shells (3s and 3p) for the elements in this period. Therefore, this period has (2×1 + 3×2) = 8 chemical elements beginning with another alkali metal sodium ending with the inert gas argon.
Fourth Period
There are three sub-shells (4s, 4p, and 3d) for the elements in this period. Therefore, this period has (2×1 + 3×2 + 5×2) = 18 chemical elements beginning with an alkali metal potassium and ending with an inert gas krypton.
Fifth Period
There are also three sub-shells (5s, 5p, and 4d) for the elements in this period. Therefore, this period has (2×1 + 3×2 + 5×2) = 18 chemical elements beginning with the alkali metal rubidium and ending with inert gas xenon. It contains eight typical elements and ten transition metals.
Sixth Period
There are four sub-shells (6s, 6p, 5d, and 4f) for the elements in this period. Therefore, this period has (2×1 + 3×2 + 5×2 + 7×2) = 32 chemical elements beginning with the alkali metal cesium and ending with an inert gas radon.
The sixth period contains eight typical elements, ten transition metals, and fourteen lanthanides or rare earth elements. The lanthanides (lanthanum to ytterbium) are placed in the lower position of the periodic table.
Seventh Period
There are also four sub-shells (7s, 7p, 6d, and 5f) for the elements in this period. Therefore, this period has (2×1 + 3×2 + 5×2 + 7×2) = 32 chemical elements beginning with the radioactive francium and ending with oganesson.
Most of the elements in this period are radioactive. The actinides (actinium to nobelium) are placed in the lower position of the periodic table.
Groups in Periodic Table of Elements
The vertical columns are called groups for periodic table elements. The modern or long form of the periodic table contains eighteen (18) groups where chemical elements are arranged.
- Groups 1 and 2, and groups 13 to 17 contain the typical chemical elements of the period table. All the elements of a particular group of normal elements have the same number of valence shell electrons in their outer quantum shell.
- Group 3 to Group 12 elements are called transition metals.
- Group 18 contains inert gases starting with helium and ending with oganesson.
In the modern periodic table, the elements on the left side are metals and the right side are nonmetals. The transition elements are placed between the metals and nonmetals of the periodic table.
Periodic Table of Elements List
The list of chemical elements arranged in groups and periods of the periodic table are listed below in the table,
| Group | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Period 1 | |||||||||||||||||||
| 2 | |||||||||||||||||||
| 3 | |||||||||||||||||||
| 4 | |||||||||||||||||||
| 5 | |||||||||||||||||||
| 6 | |||||||||||||||||||
| 7 | |||||||||||||||||||