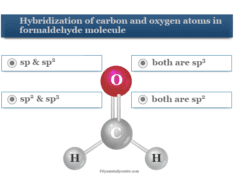
Atomic structure practice test
Chemistry quiz 4 or atomic structure practice test contains 10 questions answers for 9, 10, 11, and 12 grade chemistry students to help in exams like NEET, KVPY, etc. Therefore, Start the below online quiz to evaluate yourself.