Chemical Bonding in Chemistry
Chemical bonding or chemical bond in learning chemistry is the different types of forces that bind together two common atoms or groups of atoms forming an aggregate of ions, molecules, or compounds by lowering energy. Ionic bond, covalent bonding, metallic bonding, and hydrogen bonding are common types of bonds in chemistry or chemical science. Every atom in a chemical compound has a saturation capacity of the electron around the nucleus for the formation of the chemical bond. Valency is mostly used for the saturation capacity of periodic table elements for chemical bonding. The definition and formation of ionic, covalent, metallic, and hydrogen bonding or bonds explain the different types of properties like polarity, dipole moment, electric polarization, oxidation number or state, etc.
Types of Chemical Bonds in Chemistry
Ionic, covalent, metallic, and hydrogen bonds are common types of chemical bonding in chemistry or chemical science. However, there will be many examples of chemical bonding whose properties and structure indicate the bond of intermediate types. Such a type of chemical bonding is called a coordinate covalent bond.
Compounds and Chemical Bonds
The fundamental questions in learning chemistry, since the beginning what forces are responsible for the formation of bonds in chemical compounds?
After nearly a century of confusion, Kekule, Van’t Hoff, Le Bel, Lewis, and London in the nineteenth century defined ionic and covalent chemical bonds or bonding. It was easily realized that the number of atoms or groups of atoms combines to form the ions or molecules.
Ionic Chemical Bond
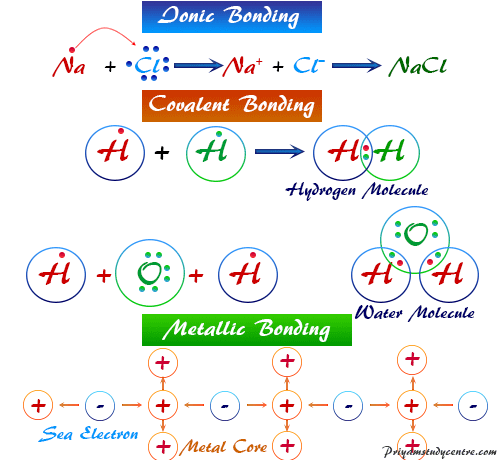
The electrostatic forces bind together oppositely charged ions in chemical compounds responsible for the formation of ionic bonds. Therefore, the ionic bonding in the molecule is formed by the transfer of electron or electrons from an electropositive metal to an electronegative non-metal atom.
- Electropositive atoms of chemical elements tend to lose one or more electrons to form positive ions.
- However, the electronegative atom of chemical elements tends to gain these electrons to form negative ions.
The mutual electrostatic attraction between such positive and negative ions establishes the formation of an ionic bond in chemical compounds.
Examples of Compounds
Sodium chloride (NaCl), potassium chloride (KCl), magnesium sulfide (MgS), calcium chloride (CaCl2), and calcium oxide (CaO) are examples of common compounds formed by ionic bonding.
The formation of sodium chloride and calcium oxide is given below the picture,
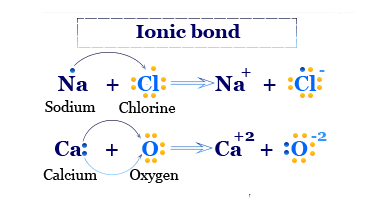
Formation of Ionic Bonding
Every halogen atom has seven electrons in its outermost orbital. Therefore, the halogen atom gains one electron by chemical bonding to attain the stable electronic configuration of the next noble gas molecule.
On the other hand, alkali metal with very low ionization energy has one electron in the outermost orbitals. Therefore, alkali metal tries to lose this electron for the formation of chemical bonding to form a stable noble gas electronic configuration.
Ionic Bonding in Sodium Chloride
Formation of NaCl, the sodium atom loses one electron to form a positively charged Na+ ion. But the chlorine atom gains one electron to form a negatively charged Cl− ion.
Na → Na+ + e
Cl + e → Cl−
Na+ + Cl− → NaCl
These two oppositely charged ions are associated or bonded together by electrostatic attraction to form a sodium chloride crystalline solid.
Crystallographic studies of sodium chloride show that there is no discrete sodium chloride molecule in nature. Hence in sodium chloride crystal lattice, each potassium ion is surrounded by six chlorine atoms or vice versa.
Properties of Ionic Compounds
- The cations and anions in ionic compounds are held together by electrostatic forces of attraction.
- These compounds are polar and soluble in polar solvents like water, ammonia, etc.
- Ionic compounds are ionizable in solution or a fused state.
- The solution of ionic compounds is a good conductor of electricity.
- They possess high melting points and boiling points.
- The polar linkages present in ionic compounds are also non-directional.
Covalent Chemical Bond
G.N. Lewis in 1916 first proposed the formation of chemical bonds in the molecules by atoms without any transference of electrons from one to another. Lewis suggested that the union of atoms by bonding in molecules,
- Hydrogen, nitrogen, oxygen, chlorine, etc
- Most of the organic compounds like hydrocarbon, alcohols, organic acids, etc.
These types of bonds are formed by the sharing of electrons pair between the atoms. In such a way, the participating atoms complete their octet or form a stable noble gas electronic configuration.
Formation of Covalent Bond
For example, the carbon atom has four electrons in the outermost shell. Therefore, the carbon atom needs four electrons to complete the octet. If these four are obtained from four chlorine atoms by common sharing, carbon tetrachloride was formed by the covalent chemical bonding.
In each bonding, the chemical atom attains its stable inert gas configuration. In the case of the hydrogen atom and carbon atom in the methane molecule, the hydrogen atom bonds with carbon by sharing electrons.
Lewis Structure for Covalent Compounds
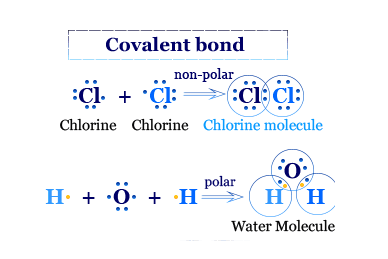
Lewis structure explains clearly the formation of covalent bonds through the sharing of electrons and also the most chemical behavior like polarity, dipole moment, or polarization of covalent compounds. However, the theory does not provide the mechanism of sharing obtained from the learning of wave mechanics.
Properties of covalent compounds
- The covalent crystal lattices are formed by weak Van der Waals forces of attraction.
- These are generally soluble in organic solvents.
- The melting and boiling points of covalent compounds are low.
- Covalent compounds are generally soft, easily fusible, and volatile in nature.
- The covalent chemical bond has directional properties.
Coordinate Covalent Bond
The sharing of electrons equal to the partner is sometimes not common in the definition of covalent chemical bonds.
For the formation of the bond between boron trichloride and ammonia, both the electrons come from ammonia. Hence such types of chemical bonding are examples of coordinate covalent bonds.
Here ammonia acts as Lewis acid, and boron trifluoride acts as Lewis base,
NH3 + BF3 → F3B←:NH3.
Metallic Bonding
Metals are a good conductor of electricity. The formation of the metallic bonds given crystalline solid with high coordination numbers of 12 or 14. The covalent and ionic chemical bonds are not possible in metals.
The atoms in a metal are identical they can not show ionic properties because an ionic bond is formed between two different atoms of chemical elements.
Covalent bonds are also not possible for metals. In covalent compounds, much weak Van der Waals force acts between the two bonding chemical atoms. It can not explain the rigidity of the metal atom.
The metallic chemical bonding may be the collection of positive atomic cores and mobile electrons in the electron sea model. The chemical force that binds the metal and mobile electrons is called a metallic bond. The electron sea model in metallic bonding can mostly explain the conductance and conduction of heat in metal compounds.
Metallic Bonding Properties
- Under the influence of the electric field, the electrons of the metal move through the lattice. Thus metals are the conductor of electricity.
- Heat conduction appears due to motion in electrons. The higher chemical energy transfers some energy to mobile electrons for the formation of metallic bonding. But these electrons move from one atomic core to another. Therefore, metal is a good conductor of heat.
- Although most of the chemical properties of the metal can be explained by metallic bonding the heat capacity of metals is difficult to explain by the electron sea model definition.
Chemical Bonding of Atoms
We can easily learn that the ions of chemical elements form an ionic bonding by electrostatic force of attraction. However, it is not clear which forces hold the two atoms in the covalent bonds.
After the development of quantum mechanics, valence bond and molecular orbital theory explain better the chemical nature of covalent bonding. Another type of bonding like hydrogen bond used for explaining some common physical properties of the compound.