Mass Defect and Nuclear Binding Energy
Nuclear Binding energy and mass defect can be calculated from the expected mass of any nucleus from the knowledge of the nuclear composition like masses of proton and neutron particles. In chemistry, such binding energy calculation formula shows that the calculated mass of a nucleus is always greater than the experimentally determined mass. This difference is called the mass defect, mass deficit, or mass decrement of an atom in nuclear chemistry and such calculation is used to find out binding energy. Binding energies vary greatly with the bound particles or the composition of the nucleus of periodic table elements. An idea of the relative stability of the stable nuclei of different chemical elements can be obtained by the plot of the binding energy per nucleon against the mass number of elements.
Mass Defect Formula
Let an atom have mass number = A and atomic number = Z. The atom contains Z number of proton and (A − Z) number of neutron particles in the nucleus.
Therefore, the mass of the atom is expected to,
(A − Z)mn + Zmp + Zme.
However, the isotopic mass (M) is usually less than the theoretically calculated value. Therefore the difference is called mass defect equal to the value,
D = (A − Z)mn + Zmp + Zme − M
= (A − Z)mn + ZmH – M
Where mn = mass of the neutron,
mp = mass of the proton,
me = mass of the electron,
Mass of hydrogen atom = mH = mp + me
Mass Defect of Helium
For example, the helium nuclei (2He4) have the following data for binding energy and mass defect calculation, the mass of two protons and neutrons = 2.01456 and 2.01743 amu respectively.
Therefore, the total mass,
= (2.01456 + 2.01743)
= 4.03190 amu
However, the actual mass of helium nuclei (2He4) = 4.0015 amu. Hence (4.03190 – 4.00150) = 0.03040 amu is the mass defect of helium. This mass defect is converted or transformed into energy by the Einstein relativity equation in physics or nuclear chemistry.
Binding Energy of Nucleus Formula
According to the definition of binding energy, the energy required to hold together the individual protons and neutrons of a single nucleus. It is calculated from the loss of mass or mass defect.
The mass loss is converted into energy according to the law of mass energy conservation with the Einstein equation,
E = mc2
1 amu mass produces 1.492 × 10−3 erg or 931 MeV of energy.
The energy formula derived from the Einstein mass-energy equation is responsible for holding the constituent particles together. Therefore, the calculated binding energy per nucleus in MeV units = 931 × D.
For example, the mass defect in the deuterium atom,
= mp + mn + me − m
= 0.00293.
Therefore, EB = 931 × 0.00293
= 2.72 MeV
Average Binding Energy Per Nucleon
The mass defect for the helium atom = 0.03040 amu
EB = 0.03040 × 931 MeV = 28.3 MeV
However, the helium nucleus contains a total of four nuclear particles or nucleons. The average binding energy per particle or nucleon = 1.36 MeV but the mean value = 28.3 MeV/4 = 7.07 MeV.
| Atom | Binding Energy (MeV) | Mean EB per Nucleon |
| 7Li | 39.27 | 5.61 |
| 12C | 92.30 | 7.69 |
| 20Ne | 160.00 | 8.00 |
| 209Bi | 1630.00 | 7.80 |
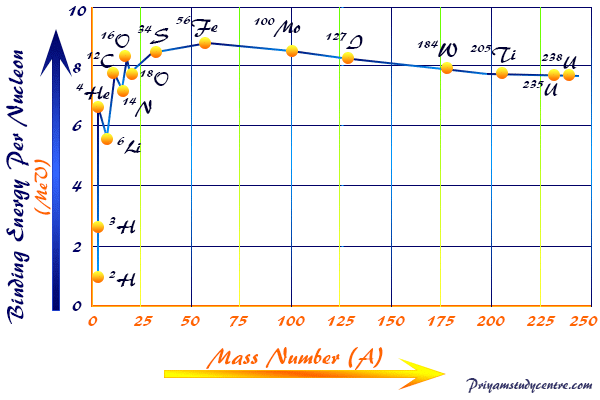
The mean binding energy formula per particle (proton, and neutron) or nucleons is different in different atoms expressed above the table laying between the values 7.5 to 8.5 plotted against the atomic number of the curve or graph.
The plot shows that the binding energy per nucleon rises sharply from the isotopes of hydrogen to the next heavier chemical elements and produces a large amount of energy by binding or fusing together or nuclear fusion.