Ideal Gas Law Problems and Solutions
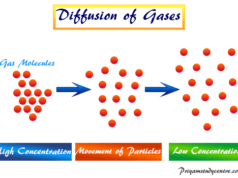
Ideal gas law problems and solutions contain online quiz questions and answers for school college grades or class chemistry students to help in examinations. Among the solid, liquid, and gaseous states of matter, only gas molecules allow for a simple comparative study of pressure, volume, and temperature. Molar mass, volume, temperature or critical temperature, density, kinetic energy, specific heat, and diffusion are the basic properties of gases. The ideal gas law defines the gases that obey the ideal gas equation in all temperature and pressure. An ideal gas is a hypothetical gas consisting of identical particles of negligible volume with no intermolecular forces. Practically there is no ideal gas and the concept of the ideal gas is a theoretical one.

Before starting to solve the questions on ideal gas law, read the properties of gas molecules and different types of laws related to the gaseous states of matter. Start the test below and solve the ideal gas quiz to test your knowledge about ideal gases: