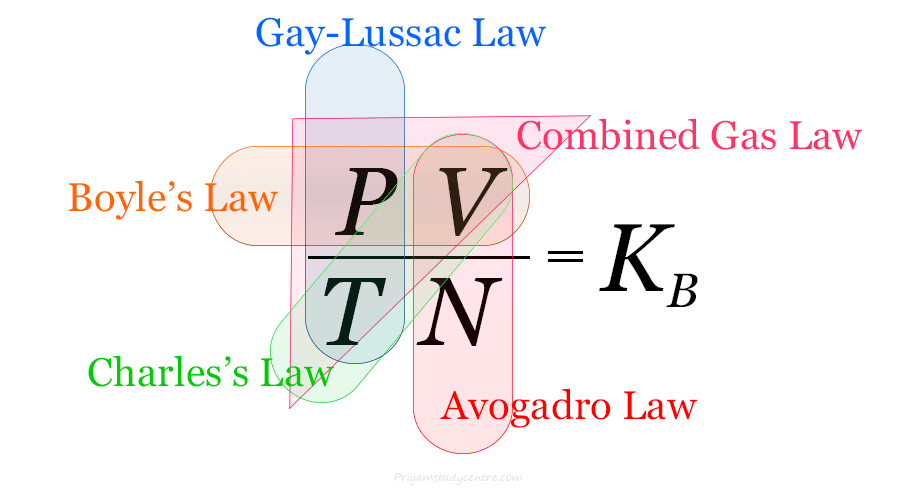Graham’s Law
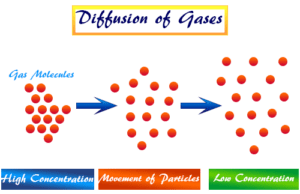
Graham's Law of Diffusion
Graham's Law of diffusion and effusion in chemistry was proposed by Scottish physical chemist Thomas Graham in 1948 to study the...
Van der Waals Equation
Van der Waals Equation of State
Van der Waals equation of state is given by modifying the ideal gas law in 1873. He put the...
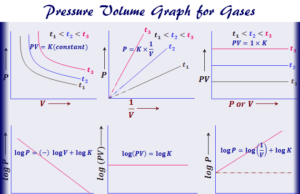
Ideal Gas Law
Ideal Gas Law Equation
Ideal gas law or perfect gas law represents the mixed relationship between pressure, volume, and temperature of hypothetical ideal gases for...
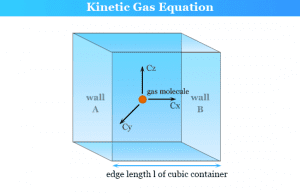
Kinetic Theory of Gases
Kinetic Molecular Theory of Gases
Kinetic molecular theory of gases and the kinetic gas equation was first developed by Bernoulli in 1738 to derive the molecular...
Ideal Gas Law Problems
Ideal Gas Law Problems and Solutions
Ideal gas law problems and solutions contain online quiz questions and answers for school college grades or class chemistry...
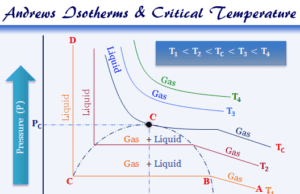
Critical Constants
Critical Constants of Gases
Critical constants like critical temperature (Tc), critical pressure (PC), and critical volume (VC) of gas in chemistry determine the condition and...
Gases
Formula of Gases
Gases are the state of matter in which molecules are far apart from each other and characterized by a lack of definite...
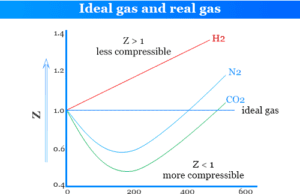
Ideal and Real Gases
Deviation of Real Gases from Ideal Behaviour
Ideal gas and real gas molecule can be compared by the ideal gas law or equation for different types...
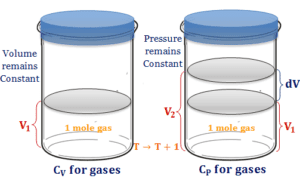
Heat Capacity Gases
Specific Heat Capacity of Gases
Specific heat capacity of gases is defined as the amount of heat required to raise the temperature of one gram of...