Benefit of Collagen Protein
Collagen is the most vital protein in mammals which forms approximately one-third of the total body protein. Collagen is a type of beneficial protein in the body that is found or uses mostly in our connective tissue of cartilage, bones, tendons, ligaments, and skin. The structure of collagen contains several amino acids which bound together to form a triple helix of the elongated fibril. Collagen protein forms 90% of the organic matrix of bones, 85% of tendons, 70% of skin, and 4% of the liver.
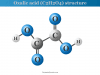
Collagen is also found in some other parts of our body such as teeth, cornea, and blood vessels. The function or role of collagenous substances is not only limited to forming a framework or structure of our body but also participates in the clotting of blood.
The production of collagen protein gradually decreases at increasing age. The production most quickly drops due to smoke, excess sun exposure, take excess alcohol, and lack of sleep and exercise. Environmental pollution can damage collagenous fibers and reduce their thickness and strength. Therefore, collagen may cause wrinkles on your skin surface.
Structure of Collagen Protein
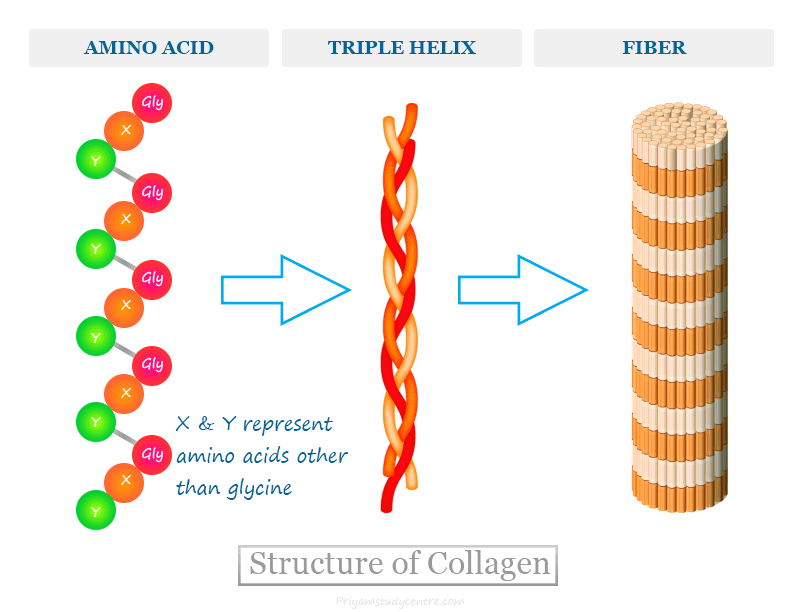
All types of collagen contain triple helical structures which may occur throughout the molecule or only a part of the molecule.
Type I mature collagen contains about 1000 amino acids that process a triple helical structure throughout the molecule. It is composed of three similar polypeptide chains twisted around each other to form a rod-like molecule.
The triple helical conformation of collagen protein can be stabilized by an extensive network of hydrogen bonding, covalent cross-links, electrostatic or ionic and hydrophobic interactions, and Van der Waals forces.
The amino acid composition in collagen molecules is unique. Approximately one-third of the amino acids present in this protein are glycine. Therefore, every third amino acid in the collagen molecule is glycine. Hence repetitive amino acid sequences in collagen molecules have been represented as (Gly-X-Y)n, where X and Y represented amino acids other than glycine.
Collagen may be regarded as a polymer of the glycine-led tripeptide found mostly in our connective tissue of bones and skin. Proline and hydroxyproline are the other two amino acids that are present in large quantities. These two amino acids confer the rigidity of collagen molecules.
Collagen Synthesis
Collagen production or synthesis occurs naturally in fibroblasts and related cells such as osteoblasts in bones, chondroblasts in cartilage, and odontoblasts in teeth. The synthesis occurs both intracellularly and extracellularly.
Inside the Cell
- Transcription of mRNA: At the beginning of collagen synthesis, a specific mRNA sequence is transcribed to form a particular alpha peptide chain.
- Preprocollagen formation: mRNA present in the cell moves into the cytoplasm and interacts with ribosomes for translation. The precursor form of collagen, namely pre-procollagen is synthesized in the ribosomes. Preprocollagen contains a single peptide chain that directs the protein to reach the endoplasmic reticulum (ER).
- Procollagen formation: In the endoplasmic reticulum, the single peptide cleaved to form procollagen. Three modifications are needed for the formation of the alpha peptide from pre-pro-peptide. It occurs by hydroxylation, glycosylation, and disulfide bond formation. In this synthesis pathway, three of the hydroxylated and glycosylated propeptides twist to form a triple helix structure of procollagen.
- Golgi apparatus modification: The procollagen goes through one last post-translational modification in the Golgi apparatus before being secreted in the extracellular medium.
Outside the Cell
- Formation of tropocollagen: Once outside the cell, membrane-bound enzymes amino-proteinase and carboxypeptidase remove the terminal amino acids of the procollagen molecule. The left molecule is called tropocollagen.
- Collagen fibril formation: An extracellular copper-dependent enzyme, lysyl oxidase acts on lysines and hydroxylysines producing aldehyde groups. These may eventually undergo covalent bonding between tropocollagen molecules to form collagen molecules. The polymer form in this synthesis pathway is called collagen fibril.
Types of Collagen Protein
As of 2011, 28 types of human collagen have been identified and described. These are divided into several groups according to their structure. All of these types contain at least one triple helix structure.
Collagen is not a single homogeneous protein but a group of structurally related and genetically distinct proteins found widely in the human body. The types of collagens are numbered by Roman numerals I, II, III, IV, V, VI, VII, VIII, IX, X, etc.
The different types can perform specialized biological functions in tissues. The five most common types of collagens are,
- Type I: Type I collagen is the main component of the organic part of our bone and is also found widely in the skin, tendon, vasculature, organs
- Type II: It is the main collagenous component of cartilage
- Type III: It is the main component of reticular fibers
- Type IV: It forms basal lamina and an epithelium-secreted layer of the basement membrane in the human body
- Type V: It is found mainly in cell surfaces, hair, and placenta
Collagen Protein Sources
Collagen is naturally found in animal-based foods such as meat and fish that contain connective tissue. However, a variety of animal and plant foods contain materials that have been used for the production of collagen in our bodies.
It is produced naturally in your body by combining various amino acids that are found in various foods. For the production of collagen in your body, we need:
- Proline: Proline is an important amino acid for human health that has been found in egg whites, dairy, cabbage, mushrooms, and asparagus.
- Glycine: It is found in pork skin, chicken skin, gelatin, and a variety of other protein-based foods.
- Vitamin C: It is found mostly in citrus fruits and bell peppers.
- Zinc: The major sources of zinc for human health are beef, lamb, pork, shellfish, chickpeas, lentils, beans, milk, cheese, and various nuts and seeds.
- Copper: It is an important mineral for the human body that is found in organ meats, cocoa powder, cashews, sesame seeds, and lentils.
The strength of the collagen fibers is contributed by the covalent cross-links formed between lysine and hydroxylysine residues. In older people, the collagen-containing tissues in the skin and blood vessels become less elastic and stiffer. Therefore, it causes health complications in older people.
Collagen Proteins Function
The name collagen comes from the Greek word kólla meaning glue, and the suffix -gen denoting producing. It is the most abundant protein in mammals containing one-third of the total body protein. They are the group of structurally related and genetically distinct proteins found in mammals.
It makes various critical parts of our bodies. Therefore, it is not only beneficial but essential to maintaining our good health. However, scientists are still debating the beneficial effects of collagen powder and related supplements.
Cardiac Skeleton of Heart
Collagen is the most important component of the cardiac skeleton of our four heart valve rings. They are histologically, elastically, and uniquely bound to cardiac muscle.
The cardiac skeleton provides a great amount of structural and functional support for the heart’s valves by enabling them to stay open. It also provides a point of attachment for the valves to the wall of the heart.
The collagenous structure that divides the upper chambers of the heart from the lower chambers is an impermeable membrane. It excludes both blood and electrical impulses through typical physiological means.
Collagen for Bones
Your bones become less dense and more brittle when you are aged. Therefore, it breaks more easily and takes longer to heal. Some research shows that daily collagen powder or supplements may increase our bone density.
Role of Collagen in Skin
Collagen may help to maintain your skin elasticity and hydration. In older people, collagen powder and supplements may have been shown to improve skin hydration and elasticity.
The strength of the collagen fibers can be contributed by the covalent cross-links formed between lysine and hydroxylysine residue. The degree of cross-linking increases with age. Therefore, in older people, collagenous tissues present in the skin and blood vessels become less elastic and stiffer which causes various health complications.
Wound Healing
Collagen is one of the most important natural resources and components that can benefit all stages of wound healing. It is a natural product which resistant to bacteria and is used for natural wound dressing.
It helps to keep the wound sterile because of its natural ability to fight against infection. When it is used for burn dressing, healthy granulation tissue is able to form very quickly over the burn. Therefore, it helps to rapidly recover healing areas.
It performs the following four biological functions during wound healing.
- Collagen fibers help to guide fibroblasts in our body that migrate along a connective tissue matrix.
- Due to the large surface area, collagenous fibers can attract fibrogenic cells which help in wound healing.
- Due to the presence of certain neutral salt molecules, it can act as a nucleating agent causing the formation of fibrillar structures.
- It can interact with blood platelets to form a hemostatic plug.
For Brain Repair
A high level of this protein may help to maintain your brain health because collagen is pure protein. Therefore, your brain needs a lot of this protein and its deficiency clearly contributes to brain degeneration and Alzheimer’s disease.
Various scientists suggest that a certain type, collagen VI may protect brain cells against amyloid-beta proteins. They are widely thought to cause Alzheimer’s disease.
Collagen is hydrogel polymers that have the capacity to store large quantities of water molecules. Hydrogels can repair the central nervous system (CNS) because they can be conveniently placed into soft nervous tissue due to their matching mechanical properties. It is the best choice for CNS or brain repair because it is highly biocompatible, biodegradable, and non-toxic.
Collagen for Hair
It provides amino acids that can be used to build hair which is primarily made up of protein keratin. Your body may use several amino acids to build keratin. Some of these amino acids can be found in collagenous products.
Therefore, when you consume collagen and other proteins, your body breaks them down into amino acids that are used to build new proteins and biological compounds. Proline is one of the main components of keratin that is obtained from collagen. Therefore, consuming proline-rich collagen may need to improve your hair health.
Uses of Collagen in the Body
Collagen has very diverse applications in medicine, the food industry, and scientific research. It is used widely in bone and cartilage reconstruction, wound dressing, tissue engineering, drug/gene delivery, and cell encapsulation systems.
- Medical industry: It is an important component of cosmetic surgery and burns surgery for artificial skin substitution. Every year several metric tons of collagenous products are used for medical applications.
- Biological laboratory: Collagenous substances are used for studying cell culture, studying cell behavior, and cellular interactions with the extracellular environment. It may also be widely used in bioink for 3D bioprinting and biofabrication of 3D tissue.
- Food industry: The degraded product gelatin is used widely in many foods. Besides food, gelatin has been widely used in the medicinal industry, cosmetic industry, and photographic industry.
- Cosmetics: It is used in dietary supplements and cosmetics for potential remedies against the aging process. It is widely used in dermal fillers for aesthetic correction of wrinkles and skin aging.
- Glue Production: It is the oldest glue in the world, which was used by Egyptians about 4,000 years ago, and Native Americans used it about 1,500 years ago.
Collagen Associated Disorders
The biosynthesis of collagen is a complex process that involves at least 30 genes in humans and about 8 post-translational modifications. Therefore, synthesis errors can present in this biosynthesis. The most common collagen-associated health disorders or diseases are
- Scurvy
- Osteogenesis Imperfecta (OI)
- Alport syndrome
- Ehlers-Danlos Syndrome (EDS)
- Epidermolysis bullosa
- Lathyrism
Scurvy
Scurvy is a disease associated with the deficiency of vitamin C (ascorbic acid) which is required for the post-translational modification of procollagen. Therefore, it is not a genetic disease but is related to the improper formation of this protein.
Osteogenesis Imperfecta (OI)
Osteogenesis Imperfecta is a genetic disorder of connective tissues that affects the bones making them weak and easily breakable. It is a disorder that is caused by an abnormality in the synthesis or processing of type I collagen.
Alport syndrome
Alport syndrome is a disorder that is caused due to a defect in the formation of type IV collagen fibers found in the basement membrane of capillaries and the glomeruli of nephrons in the kidney. It damages the tiny blood vessels in your kidneys and leads to causes kidney disease and kidney failure.
Ehlers-Danlos Syndrome (EDS)
Ehlers-Danlos Syndrome (EDS) is a group of inherited connective tissue disorders usually caused by genes that alter the structure, production, or processing of proteins that interact with collagen.
It provides structure and strength to connective tissue. Therefore, a defect in collagen formation can weaken connective tissue in the skin, bones, blood vessels, and other organs of the human body which causes disorders like Ehlers-Danlos Syndrome (EDS).
Epidermolysis Bullosa
Epidermolysis bullosa (EB) is a group of rare medical conditions where victims exhibit skin breaks and blisters formation even for minor trauma. It may be caused by to loss or diminished function of type VII collagen.
Epidermolysis bullosa is a disorder that leads to the weakening of the structural architecture of the dermal–epidermal junction (DEJ) and mucosal membranes in humans.
Lathyrism
Lathyrism is a disease of bone deformities caused due to the toxic effects of β-oxalyl aminoalanine found in Kesari dal. The compound obtained from Kesari dal may interfere with the cross-linking of lysine amino acids in this protein.
Frequently Asked Questions
How to increase collagen in skin naturally?
Eating foods that contain vitamin C and amino acids such as glycine, proline, and hydroxyproline can help to boost collagen production. Avoiding smoking, limiting caffeine intake, and protecting the skin from sunlight may help preserve such protein on the skin.
Where is collagen found?
It is naturally found in animal-based foods such as fish, poultry, meat, eggs, and dairy. However, a variety of both animal and plant foods contain materials that use for the production of collagen in our bodies.
What does collagen do?
It is a major component of connective tissue that gives strength, support, and shape to the tissues. It contributes to the proper alignment of cells which helps in cell proliferation and their differentiation into different tissues and organs.
In blood vessels, it helps for the formation of thrombus in our body.
What vitamin promotes collagen synthesis?
Vitamin C promotes the biochemical synthesis of the protein collagen. It plays an important role in the conversion of such protein from protocollagen.
Vitamin C is a cofactor for enzymes prolyl hydroxylase and lysyl hydroxylase. These two enzymes are involved in the hydroxylation of proline and lysine. Hydroxyproline and hydroxylysine are essential for cross-linking and strengthing this fiber. Therefore, vitamin C can help to promote collagen synthesis in your body and maintain normal connecting tissues of skin and uses for wound healing processes.
What is type Ⅰ collagen?
It is the most abundant type of protein found in the human body that forms collagenous fibers. Type I collagen is found in the various parts of the human body such as scar tissue, dermis, bone, tendons, ligaments, and the endomysium of myofibrils.