Supercritical Fluid Chromatography (SFC)
Supercritical fluid chromatography (SFC) in analytical chemistry is a form of high performance liquid chromatography (HLPC) instrument that is used for the separation of drugs, herbicides, natural products, and food products. It is a normal phase chromatography where we use a supercritical fluid such as carbon dioxide, ammonia, water, methanol, n-butane, n-propanol, etc. Carbon dioxide is the best fluid in supercritical fluid chromatography due to compatibility with the FID detector and low critical temperature (Tc). It was discovered in 1985 by hybridization of HPLC and GLC with the advantages of both. The application of supercritical fluid chromatography is most useful because it permits the separation and determination of compounds which is not separated by HPLC or GLC.
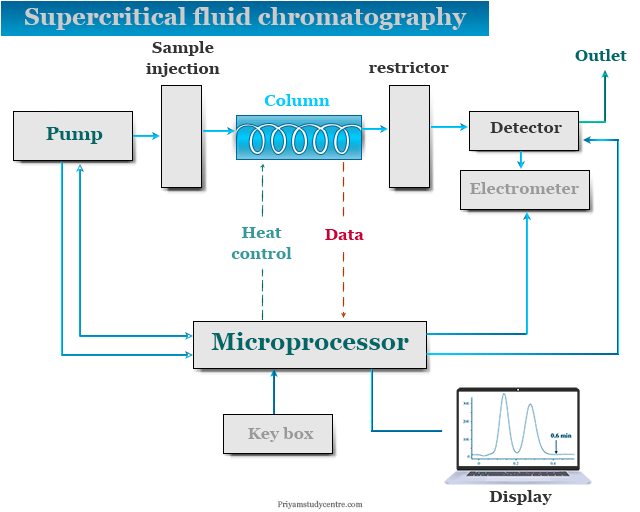
Supercritical Fluid Chromatography Instrumentation
Commercial instrumentation of supercritical fluid chromatography technique is given below the picture,
The most important components of the SFC instrument are,
- Pressure regulator
- Stationary phase
- Mobile phase
- Detectors
Pressure Regulator
The pressure regulator is an important component in the SFC instrument. It affects the density of the mobile phase. When density increases, the solvent power of the mobile phase also increases. It reduces the elution time of the SFC instrument.
For example, if the pressure of carbon dioxide molecules reduces from 70 to 90 atmospheres, elution time reduces from 25 to 5 minutes. Therefore, pressure programming is an important component of the supercritical fluid chromatography principle.
SFC Column
We used an open or closed column in the SFC technique. The columns are 10 to 20 m in length and 0.05 to 0.10 mm thick. The column used in the SFC technique is similar to those used in high-performance liquid chromatography (HPLC) or other column chromatography.
SFC Mobile Phase
The commonly used mobile phase for supercritical fluid chromatography is carbon dioxide (CO2). It is the best solvent for a variety of organic compounds. It is used widely in SFC instrument because CO2 transmit ultraviolet visible spectrum, is nontoxic, and is cheap.
Sometimes polar organic modifiers like methanol are used on it. We used also ethane, pentane, nitrous oxide, dichlorofluoromethane, ether, and ammonia modifiers.
Detector
The flame ionization detector (FID) is used in the SFC instrument but cannot be used in HPLC. It can be hyphenated with a mass spectrometer. Other detectors like UV, IR, and fluorescence can also be used in SFC instrumentation.
Supercritical Fluid Chromatography Applications
- It is applied for the separation of a variety of materials such as drugs, herbicides, pesticides, and natural food products.
- SFC technique is especially useful for the separation of a series of dimethylpolysiloxane and oligomers. The separation of oligomers in nonionic surfactants like triton X-100 can be done by carbon dioxide mobile phase with 1% methanol as a modifier.
- The separation of compounds with molecular weight from 400 to 700 daltons can be done by carbon dioxide as a mobile phase.
- Polyaromatic hydrocarbon can be easily separated by supercritical fluid chromatography.