Green Chemistry Principles
Green chemistry or sustainable chemistry is the field of chemistry and chemical engineering which can be used to design, develop, and implement chemical products and processes. The principles of green chemistry were developed by scientists and engineers to protect our environment from harmful waste, save energy, and discover how to replace hazardous substances. The 12 (twelve) principles and research on green chemistry focus on the environmental importance of chemistry or chemical substances in daily life, reducing the use of nonrenewable natural resources, and technological solutions for environmental pollution. On the other hand, environmental chemistry only focuses on polluting substances in our environment.
What is Green Chemistry?
Sets of green chemistry principles that reduce environmental hazards and health problems were published by Paul Anastas and John C. Warner in 1998. The twelve (12) principles of green chemistry can be used for the development of research and journals. The principal aim of green chemistry concepts is,
- Using less hazardous reactants, products, and solvents for the synthesis of daily used chemicals.
- Designing processes that can maximize the use of raw material or produce less amount of waste.
- Designing such type of synthesis process which can be converted maximum reactants into desired products.
- Using renewable energy sources to minimize various types of pollution such as air pollution, water pollution, greenhouse effect, global warming, etc.
- Using energy-efficient processes to save energy for our future.
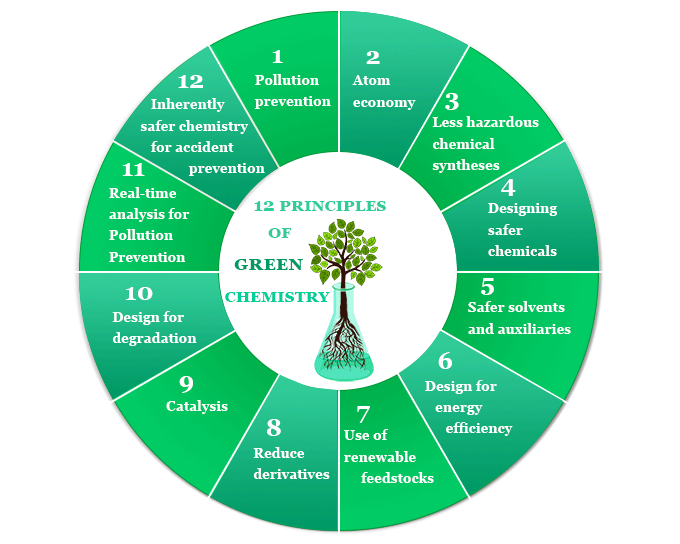
12 Principles of Green Chemistry
The twelve (12) principles of green chemistry or sustainable chemistry are,
- Pollution prevention
- Atom economy
- Less hazardous chemical syntheses
- Designing safer chemicals
- Safer solvents and auxiliaries
- Design for energy efficiency
- Use of renewable feedstocks
- Reduce derivatives
- Catalysis
- Design for degradation
- Real-time analysis for pollution prevention
- Inherently safer chemistry for accident prevention
Pollution Prevention
Pollution prevention is any process that can be reduced, eliminated, or prevented from polluting substances in our environment. Reducing waste is better than treating or cleaning up waste.
Atom Economy
Atom economy in green chemistry is the conversion efficiency of a chemical process in terms of all atoms involved in the chemical reactions and less harmful products are produced. It is designed to generate less waste in any chemical process.
Less Hazardous Chemical Syntheses
Using synthesis methods to create less hazardous or non-toxic substances for humans and other living organisms is the main aim of this principle.
Designing safer chemicals
Many toxic chemicals used in our daily lives have effects on human health and the environment. Different policies and systems are governed to ban these chemicals. Removing the chemicals once they are already in the marketplace can be difficult, time-consuming, and costly. Therefore, this principle of green chemistry is implemented to identify problematic chemicals and design safer chemicals that achieve their desired function.
Safer Solvents and Auxiliaries
This principle of green chemistry is suggested to avoid the use of auxiliary substances wherever possible. It also promotes the use of safer solvents for carrying out chemical processes.
Design for Energy Efficiency
This principle is designed to minimize the energy requirement and economic impacts. Therefore, the chemical process should be conducted at ambient temperature and pressure whenever possible.
Use of Renewable Feedstocks
The use of renewable feedstock is one of the more important materials of modern green chemistry than non-renewable fossil resources. Most of the energy supplied to the world comes from non-renewable fossil resources. These fossil resources are not renewed after a time interval but energy demand in our world increases day by day. In daily life, the primary importance of this sustainable chemistry principle or research is to produce cost-effective alternatives derived from renewable resources.
Reduce Derivatives
This principle of green chemistry suggests that unnecessary derivatives or steps should be minimized or avoided during any chemical process if possible. Such derivatives or additional steps require additional reagents or generate toxic waste.
Catalysis
Catalysis is one of the fundamental principles of green chemistry that design or eliminate environmentally hazardous substances or toxic waste. It is used in the chemical processes for achieving dual goals like environmental protection and economic benefit. The economic benefits of green chemistry improve the corporate bottom line. A wide amount of production costs are decreased through the use of green chemistry.
If less waste is generated, environmental pollution can go down. Therefore, the treatment and disposal become unnecessary. Catalysis in greener technologies is also beneficial for human health and our environment.
Design for Degradation
The design for degradation principle in sustainable chemistry is designed for the degradation of chemical products after their function is completed. Therefore, chemical products obtained from different processes do not pollute the environment and they should break down into non-harmful products.
Real-time Analysis for Pollution Prevention
Analytical techniques are used for analyzing different chemical substances in real time to prevent pollution. Real-time analysis can monitor and control harmful substances before they form.
Inherently Safer Chemistry for Accident Prevention
The substances obtained from the different chemical processes are safer to use. This principle of green chemistry is used to minimize accidental risks such as explosions, fires, health problems, etc.
Applications of Green Chemistry
Green chemistry or sustainable chemistry is beneficial for many chemical processes. It can minimize environmental hazards and be beneficial for human health and the ecosystem. The main importance or applications of green chemistry in daily life are,
Carbon Dioxide
In green chemistry, dense phase carbon dioxide should be used for food and pharmaceutical processing. It is a safe product with minimal nutrient loss and better preservation quality. The significant amount of research on dense phase carbon dioxide suggested its effectiveness against microorganisms and enzymes.
High-pressure carbon dioxide is an efficient Boeing agent for polymer processing due to its chemical and economic advantages. It produced mainly polystyrene foam sheets. These are used for packing in our daily life. It also replaces the previously used Boeing agent CFC which can cause ozone layer depletion.
Direct Methanol Fuel Cell
The different research paper describes recent developments in the methanol-oxygen fuel cell or direct methanol fuel cells (DMFCs). It can convert the chemical energy of liquid methanol into electrical energy and produce a small amount of power over a long period of time. These fuel cells are used in different types of electronic equipment, such as notebooks, video cameras, cell phones, medical devices, etc.
Antifouling Agents
Antifouling agents or paints are the special types of coating on marine vehicles that reduce the buildup of microorganisms on marine vehicles. Through sustainable chemistry research, we can produce various non-toxic biodegradable antifouling agents. These coatings are not yet commercially available and are designed by different types of research.
Oxidative Cracking
Environment-friendly catalytic processes are important for modern society due to the ever-increasing demand for such catalytic processes. The development of highly active and selective chemical catalysts is part of green chemistry research.
The oxidative creaking was discovered through sustainable chemistry research. A special type of oxidative creaking was established from green chemistry research for the production of ethylene from higher hydrocarbons. It can save a huge amount of natural gas and reduce the emission of carbon dioxide.
Pyrocool Foam
Pyrocool foam is prepared from green chemistry research. In 1993, Pyrocool Technologies Inc. acquired the patent rights due to superior wetting and cooling properties. They are effecting effective firefighting agents for fighting wildfires and flammable liquids. It is an environmentally friendly cooling agent which minimizes several types of environmental pollution.
Antibiotic Drugs
Environmental friendly antibiotics are a major concern for various governments, regulatory agencies, and the pharmaceutical and cosmetic industries. Green chemistry research and principles design the best process for producing various important antibiotics like ampicillin, amoxicillin, etc.
Importance of Green Chemistry in Daily Life
Sustainable chemistry research and journals are beneficial for human health, the environment, the economy, and business. In daily life, the most common importance of green chemistry is,
- Less release of hazardous chemicals into air and water leads to reduced air pollution and water pollution.
- Sustainable chemistry research is involved in the production of safer or less toxic consumer products. These are beneficial for human health.
- It also potentially lowers various environmental problems such as global warming, acid rain, ozone depletion, and smog formation.
- Higher yields for chemical reactions, consuming smaller amounts of feedstock, and lowering synthetic steps in sustainable or green chemistry principles or research allow faster manufacturing of lower-cost products.