Infrared (IR) Spectroscopy
Infrared spectroscopy also called IR spectroscopy or vibrational spectroscopy is the analysis of infrared light that interacts with matter in the infrared region of the electromagnetic spectrum. An infrared spectrometer is an instrument based on absorption in the infrared region due to changes in vibrational energy. An IR spectrometer is the finest tool for the characterization of various inorganic and organic compounds. Infrared radiation is electromagnetic radiation having a wavelength range of 0.7-1000 μm and a frequency range of 14000–10 cm−1. Analysis of frequencies in infrared spectra provides a piece of information about the flexibilities of the chemical bond, bond dissociation energy, and bond length of the molecule. IR spectra can also help to identify the functional groups, isotropic mass, etc. Therefore, it is a very important tool for the structural study of molecules.
Regions of the Infrared spectrum
The infrared spectroscopy region constitutes three parts,
- Near-infrared: The near-infrared region lies approximately in the frequency range of 14000–4000 cm−1 (0.7–2.5 μm wavelength). There are few absorptions of organic molecules found in this region. Therefore, it has little use for spectroscopic purposes.
- Middle infrared: The middle infrared region lies approximately in the frequency range of 4000–400 cm−1 (2.5–25 μm wavelength). Spectroscopy in the middle infrared region is extremely useful for the study of organic compounds.
- Far infrared: The far infrared region lies approximately in the frequency range of 400–10 cm−1 (25–1000 μm wavelength). This region is also not much use for organic spectroscopy.
Types of molecular vibrations
The relative position of atoms in a molecule is not fixed but fluctuates steadily due to different kinds of vibrations. The nature of vibrations in diatomic or triatomic molecules may correlate with absorption energies. For polyatomic molecules, such vibration cannot be predicted easily due to the interaction of many centers of vibrations.
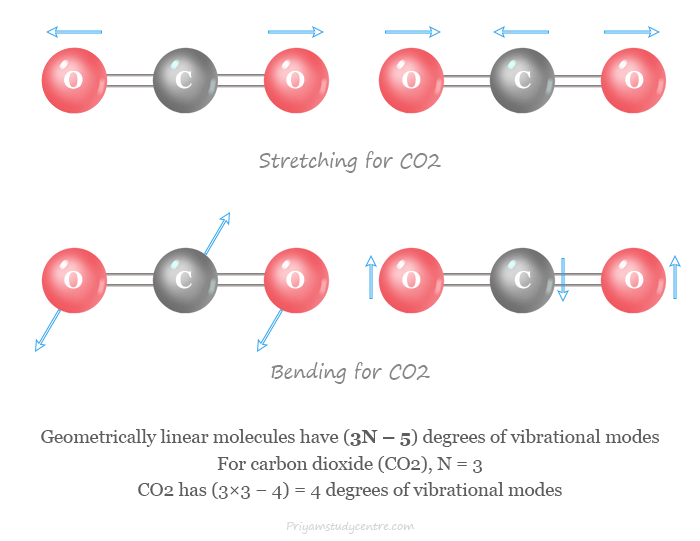
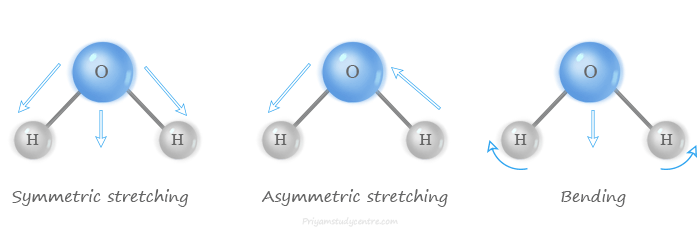
Commonly these vibrations are classified into two types such as stretching vibrations and bending vibrations.
- The stretching vibrations involve vibration along the axis of the bond connected by two atoms.
- The bending vibration changes in the angles between two bonds. They are four types namely scissoring, rocking, wagging, and twisting.
Vibrational modes in infrared spectroscopy
A molecule can vibrate in various ways and each way of vibration is called a vibrational mode. Geometrically linear molecules have 3N – 5 degrees of vibrational modes and nonlinear molecules have 3N – 6 degrees of vibrational modes, here N = number of atoms in a molecule.
- A linear carbon dioxide (CO2) molecule has three atoms. Therefore, it has 3 × 3 – 5 = 4 degrees of vibrational modes.
- The non-linear water (H2O) molecule also has three atoms but it has 3 × 3 – 6 = 3 degrees of vibrational modes.
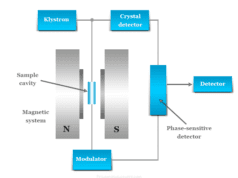
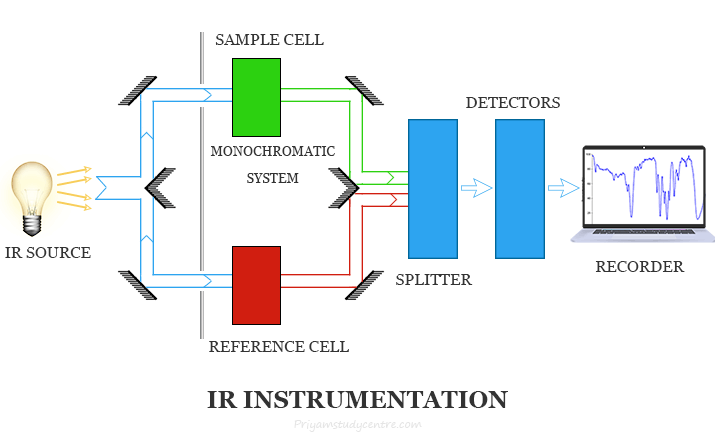
Infrared Spectroscopy Instrumentation
The instrumentation of infrared spectrometers resembles UV-visible spectrometers in many respects. The essential components of IR and UV-visible spectroscopy are similar but sources, detectors, and optical components are somewhat different from IR spectrometers.
The main components of the IR spectrometer are,
- Radiation source
- Sample cells and sampling of substances
- Monochromators
- Detectors
- Recorder
IR radiation source
The most commonly used radiation source for IR instrumentation is a Nernst or Glower’s lamp. It is made of zirconium oxides and the hallow yttrium rod with 2 mm in diameter and 30 mm in length. It is heated to 1500 to 2000 °C and gives radiation up to 7000 cm−1.
A global source is used in a few IR instruments with transmittance in the range of 5200 cm−1. The radiant energy in global sources decreases less rapidly.
Sample cells and sampling of substances
IR spectroscopy has been used for the characterization of solid, liquid, or gas samples but sample handling creates a number of problems in IR spectroscopy.
- Solid: The solid can be examined in crystalline form by dilution in a mull, an alkaline halide disc, or by spreading pure solid on a cell plate. In the nujol mull method, the sample is ground together with the mulling agent using a pestle and motor. The IR spectrum of a solid sample is often best determined as an alkali metal halide pellet.
- Liquid: Alkali metal halide cells can be used for handling liquid samples. Sodium chloride, potassium bromide, and cesium iodide are used as window martial. Whenever possible solids samples are dissolved in organic solvents like CCl4 and CS2 to prepare the solution. CCl4 is useful in the region 800-740 cm−1 while CS2 is useful in the region of 2222-1540 cm−1.
- Gas: The sample handling technique for gaseous samples is similar to that of liquid sampling.
Monochromators in IR spectroscopy
Monochromators used in IR instruments are made out of various kinds of materials. Prisms and windows are made out of glass, fuse silica, lithium fluoride, calcium fluoride, sodium chloride, silver chloride, potassium bromide, etc.
Usually, sodium chloride prism is used for measurements at 4000 to 650 cm−1 and potassium bromide prism is used for measurements at 400 cm−1.
Detectors in infrared spectroscopy
The most commonly used detector in IR instruments is a bolometer. A bolometer provides an electric signal by a change in the resistance of the metallic conductor with temperature.
It consists of a thin layer of metal connected to a thermal reservoir. PbS or PbTeO3 layer is used as coating material. The long wavelength sensitivity limit for the PbS cell is 3.5μ and for the PbTeO3 cell is 6μ.
A nonmetal detector of greater sensitivity is the photoconductive cell but it is rarely used in infrared spectroscopy.
Recorder
A recorder in IR spectroscopy is used to record the spectrum obtained from analyzed substances.
Infrared Spectrum
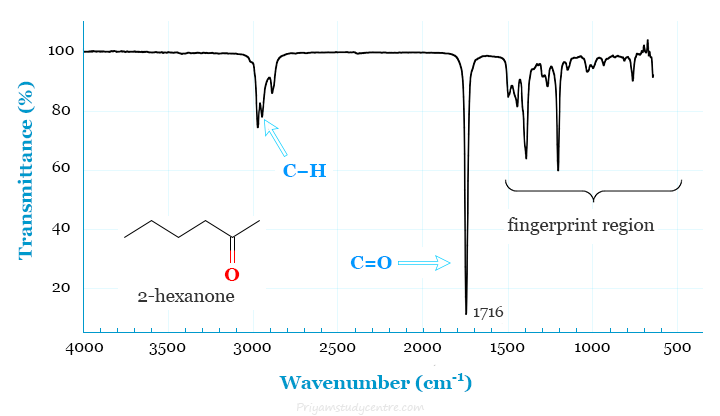
The infrared spectrum is used to identify structures of many organic and inorganic molecules because functional groups give rise to characteristic bands both in terms of intensity and frequency or wavelength. The spectrum diagram for 2-hexanone is given below the picture.
Uses of Infrared Spectroscopy
Infrared (IR) spectroscopy is a simple and reliable technique that is used widely in various fields of science to analyze organic and inorganic substances. The most common uses of IR spectrometers may include,
- Infrared spectroscopy is used by chemists to determine functional groups in organic and inorganic molecules. It measures the vibrations of atoms and based on vibrations it is possible to determine the functional groups in a molecule. Generally, stronger bonds and light atoms of a molecule will vibrate at a high stretching frequency.
- IR spectrometer is a very useful tool for catalytic research to characterize chemical catalysts and intermediates produced during catalysis reactions.
- IR spectrometer is used to identify the concentration of carbon dioxide (CO2) in our environment that causes the greenhouse effect and global warming.
- Poorly maintained vehicles exhaust two pollutants such as carbon monoxide and hydrocarbons. These two pollutants can be detected by remote sensing which contains a source of IR radiation.
- The technique can be used in forensic science to identify polymer degradation and determine the blood alcohol content of a suspected drunk vehicle driver.
- Another important application of IR spectroscopy is in the food industry to measure the concentration of various components found in different food products.
IR and Raman Spectroscopy
All modes of molecular vibration are IR active or Raman active. Therefore, Raman spectroscopy is used to examine the vibrational spectra of compounds that do not tend themselves to IR absorption spectroscopy.
The main differences between infrared and Raman spectroscopy are listed below in the table,
| Infrared spectroscopy | Raman spectroscopy |
| The IR spectrum is obtained by the absorption of light by vibrating molecules | Raman spectrum is obtained by the scattering of light by vibrating molecules. |
| The presence of a permanent dipole moment in a molecule may decide whether the IR spectra are observed or not. | Polarizability in a molecule may decide whether the IR spectra are observed or not. |
| This method is accurate and very sensitive. | This method is very accurate but not very sensitive. |
| The optical systems are made up of special crystals of alkali metal halides. | The optical systems are made up of glass or quartz. |
| Photochemical reactions do not take place in IR spectroscopy. | Sometimes photochemical reactions take place in Raman spectroscopy. |
| Homonuclear diatomic molecules are found to be IR-inactive. | Homonuclear diatomic molecules generally show the Raman spectrum. |
Fourier Transform Infrared (FTIR) Spectroscopy
Fourier transform infrared spectroscopy (FTIR) is the technique used for recording infrared spectra obtained from an analyzed solid, liquid, and gaseous sample. FTIR spectrometers are mostly used to collect high spectral resolution data in the mid and near-IR regions. The frequency of mid-IR lies between 5000 and 400 cm−1 but near-IR lies between 10,000 and 4,000 cm−1.
In the FTIR spectrometer, the infrared radiation spits into two beams. One of these beams is kept static while the other beam is moving by a mirror. These are then combined to get a modulated beam. In this modulated beam there will be more light energy or less light energy for a given wavelength. The modulated beam passes through the sample and then by a computer to give the infrared spectrum.
Advantages of FTIR spectrometer
The modern Fourier transform infrared (FTIR) spectrometer gives the same information as a simple infrared spectrometer but FTIR is best with respect to performance, speed, and sensitivity.
FTIR spectrometers are third-generation infrared spectrometers that are used widely by chemists for their analysis. The most prominent advantages of FTIR spectrometers are:
- The signal-to-noise ratio is significantly higher than the previous generation IR spectrometers.
- The accuracy of the wavenumber is high and the error is very low.
- The scan time of all frequencies is short in FTIR spectrometers.
- The resolution of the spectrum is extremely high
- The scan range is wide in FTIR spectrometers.
Application of FTIR spectrometer
The modern Fourier transform infrared (FTIR) spectrometers are widely used in organic synthesis, polymer science, petrochemical engineering, the pharmaceutical industry, and food analysis.
Fourier transform infrared spectroscopy is an important tool for nanomaterial characterization. Nano-FTIR provides a powerful tool to study polymers, 2D materials, semiconductor devices, and biomaterials such as viruses and protein complexes at nanoscale levels.
The FTIR spectral data obtained from various compounds are used for separation by gas chromatography. The use of the FTIR method is very low compared to gas chromatography-mass spectrometry (GC-MS). The combination of gas chromatography and Fourier transform infrared spectroscopy (FTIR) is only useful for identifying isomers that have identical masses.