Periodic Trends of Alkali Metals
The six elements lithium, sodium, potassium, rubidium, caesium, and francium constitute the family of alkali metals. They are placed extremely left in the periodic table. The periodic trends of alkali metals are given below the topics,
Atomic and Ionic Radius of Alkali Metals
The atomic and ionic radius of alkali metals is increasing systematically from lithium to caesium atom.
When we proceed downward on the alkali group, the electron is added to higher energy levels. It decreases the electrostatic attraction between the nucleus and valence shell electrons. This decreases electrostatic attraction and increases atomic and ionic radii.
The shielding effect also decreases the electrostatic attraction between the nucleus and valence shell electrons. With increasing shielding electrons the effective nuclear charge of the atom decreases. Therefore, nucleus and outermost electron separation increases.
Melting and Boiling Points
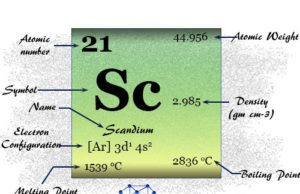
Alkali metals are soft, and low melting because only the outer ns electron participates in metallic bonding.
The melting point, boiling point, and hardness decrease with increasing atomic number. With the increasing atomic number, the size of the atom increases. It weakens the strength of metallic bonding between two alkali atoms.
Density of Alkali Metals
All the alkali metals contain a body-centered cubic crystal lattice. Therefore, the densities of these metals depend on the volume of the atom or atomic radii and atomic weight. The atomic radii and atomic weight of metals increase going down the group.
The observed densities of alkali metals increase down the group with the exception of potassium. Lithium, sodium, and potassium are the only three metals that are lower denser than water.
Ionization Energy
All the elements in the alkali metals family have one electron in the s-orbital over a noble gas core. It is easily lost to give very low ionization energy. With the increasing atomic number, the distance between the nucleus and valence s-electron increases. It leads to a steady decrease in their ionization energy.
The alkali metal contains the largest size among the elements of each period. Large size and a small charge of cation favor the formation of ionic bonding. The tendency to form ionic compounds increases down the group.
When the alkalis combine with other elements, there results in a wide difference in electronegativity. This fact leads to the formation of ionic compounds.
Oxidation Number
All the alkali metals show a +1 oxidation state or number. There is no scope for alkalies to exhibit an oxidation number of +2. To show a +2 oxidation state, alkali metals need to utilize electrons from the very stable noble gas configuration.
Standard Electrode Potential
The standard electrode potentials of alkali metals are more negative among all periodic table elements. The electrode potential values become more negative from sodium to cesium.
The alkali metal lithium may not follow the general trend of electrode potential. Lithium shows the most negative reduction potential.
The hydration energy of lithium is highest among the alkali metals due to its small size and it disrupts the structure of water molecules during electrode reactions. These two factors are quite significant in describing the reduction potential of lithium.
Flame Coloration
All the alkali metals give characteristic flame coloration due to the easy excitation of outermost electrons.
- Lithium produces crimson color
- Sodium produces a yellow color
- Potassium produces a violet color
- Rubidium gives red violet color
- Caesium gives a blue color
It led to the development of analytical chemistry for precise estimation of these metals by flame photometry and atomic absorption spectroscopy.
Chemical Reactivity of Alkali Metals
The large difference between the first and second ionization energy of these elements suggests that the preferred oxidation state of alkali metals will be +1. Therefore, the major chemistry of alkali metal is in the form of M+ ions.
The chemical reactivity of the alkali metals increases from lithium to caesium. The poor reactivity of lithium is explained by the fact that Li cannot replace the weekly acidic hydrogen from phenylacetylene.
Reaction with Oxygen
The metals react with oxygen to form respective oxides, peroxides, superoxides, and suboxides.
- Lithium reacts with oxygen to form Li2O and Li2O2.
- Sodium gives principally Na2O2.
- Other metals give superoxides like MO2.
Reaction with Water
Alkali metals react with water to form hydroxyl ions and hydrogen gas. At 25 °C, the metals react with water to create the following condition,
- lithium reacts slowly
- sodium reacts vigorously
- Potassium inflames
- Rubidium and caesium react with an explosion.
Reaction with Carbon
- Lithium and sodium react with carbon on heating to form the acetylides Li2C2 and Na2C2.
- The other alkalis give non-stoichiometric interstitial compounds.
Lithium reacts uniquely with nitrogen. At room temperature, lithium reacts slowly with nitrogen. At 400 °C, it reacts rapidly with nitrogen to give the ruby red crystalline solid Li3N.
Reaction with Liquid Ammonia
Alkali metals dissolve in liquid ammonia to give blue solutions which contain free electrons. These solutions are used as a powerful reducing agent in various organic syntheses.
Frequently Asked Questions
Which alkali metal is most reactive?
The reactivity of the alkali metals increases along the group with the increasing atomic number. With the increasing atomic number the distance between the nucleus and valence shell electron increases. Therefore, francium is the most reactive alkali metal present on the periodic table.
How many alkali metals are there in the periodic table?
There are six alkali metals present on the periodic table. The names of these metals are lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr).
Which alkali metal shows the strongest reducing properties?
Lithium is the strongest reducing agent among alkalis due to its highest electrode potential value.
Which alkali metal is radioactive?
Francium is very rare due to its extremely high radioactivity. Francium occurs in very small amounts in nature. The half-life of the longest-live isotope is 22.8 minutes.
Which alkali metal has the most metallic character?
With the increasing atomic number, the metallic character of alkali metals increases along the group. Therefore, francium is the most metallic element among alkalis.
But francium is rare in nature due to its high radioactivity. Hence the natural element that shows the highest metallic character is caesium.
What alkali metal is used in fireworks?
Among alkali metals, lithium, and sodium are used widely in fireworks. The alkali metal lithium imparts a red color to fireworks while other alkali metal sodium imparts a gold or yellow color to fireworks.