What is Spectrometer?
In learning chemistry or physics, spectroscopy is mainly concerned with the interaction of electromagnetic radiation with matter. After the interaction, there occur variations in the intensity of electromagnetic radiation over a certain range of frequencies or wavelengths.
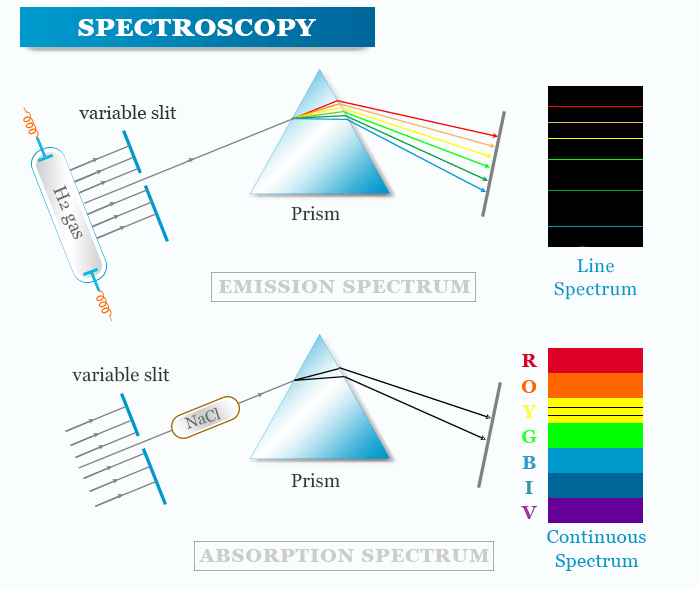
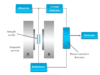
The instrument which records this variation in intensity is known as a spectrometer. Therefore, a spectrometer is a scientific instrument used to separate and measure spectral components of any substance through the analysis of its light properties.
Absorption and Emission Spectra
When a beam of electromagnetic radiation is passed through a chemical sample, a part of the radiation may be absorbed by the sample. Electromagnetic energy is transferred to the atoms or molecules present in the samples. Therefore, the particles are promoted from lower to higher energy states or exited states.
There are two common ways in which the interaction is observed,
- The sample emits radiation.
- The sample absorbed radiation from continuous sources.
Atomic Absorption Spectroscopy
Absorption spectroscopy in chemistry or physics is a spectroscopic technique that measures the absorption of radiation due to its interaction with a sample.
The study of the frequency or wavelength of absorbed radiation provides absorption spectra of the analyzing sample. Such spectra may be due to atomic or molecular absorption. It is dependent upon the physical states of matter, the environment of absorbing species, and other related factors.
When a beam of electromagnetic radiation passes through a chemical sample, a part of the radiation is absorbed by the sample. Electromagnetic energy is transferred to the atoms or molecules present in the analyzing sample and particles are promoted from lower energy states to higher energy states or exited states. Therefore, absorption involves a transition from the ground state to a higher energy state.
Atomic Emission Spectroscopy
Emission spectroscopy is a spectroscopic method that examines the wavelengths of photons emitted by atoms or molecules during their transition from an excited state to a ground state or lower energy state.
When periodic table elements or their compounds are heated on a flame or an electric arc or spark they emit energy in the form of light. Analysis of light obtained from emitted energy gives a discontinuous or line spectrum with a definite wavelength. The line spectra are useful for emission spectroscopic analysis.
Atomic emission spectroscopy can be used to determine the composition of a material because it is specific to each element of the periodic table. Astronomical spectroscopy identifies the composition of stars by analyzing the received light obtained from the star.
Fluorescence and Phosphorescence
Fluorescence and phosphorescence also called luminescence is a special kind of emission spectroscopy. They can be observed in the following ways:
In some cases, the molecules after absorbing radiation become excited but they do not lose energy very quickly.
- In such cases, the energy is re-emitted in the form of radiation with a larger wavelength than it originally absorbed. This phenomenon is called fluorescence. It occurs more rapidly than phosphorescence and completes after 10−5 seconds from the time of excitation.
- In some cases, there is a detectable time delay in re-emission. This phenomenon is called phosphorescence. It takes place over a period longer than 10−2 seconds and continues for a few minutes or even hours.
Light Scattering Spectroscopy
It is not necessary for passing radiation though the matter may be absorbed completely. A small portion of the light that passes into the matter may undergo scattered or reflected instead of being absorbed. Nephelometry and turbidimetry are based on the ability of particles to scatter light. The size shape and concentration of colloid particles and suspensions may be determined from this property.
- Elastic scattering and reflection spectroscopy determine the reflection or scattering of incident radiation by an analyzed material. Crystallography study uses the scattering of high-energy radiation to examine the arrangement of atoms in biomolecules (proteins) and solid crystals.
- Inelastic scattering is observed when there is a change in the frequency of radiation between the incident and emitted radiation. It gives rise to Raman spectroscopy. Inelastic scattering is a useful technique for environmental monitoring.
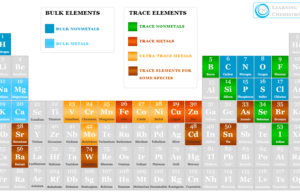
Atomic Spectroscopy
Atomic spectroscopy is the study of the electromagnetic radiation absorbed or emitted by an electron during transitions between different energy levels within an atom. When an electron of an atom is excited from one energy level to another, it either emits or absorbs light of a specific wavelength.
Atoms of different elements show distinct spectra. Therefore, atomic spectroscopy is used for the identification and quantification of an element in an analyzed sample.

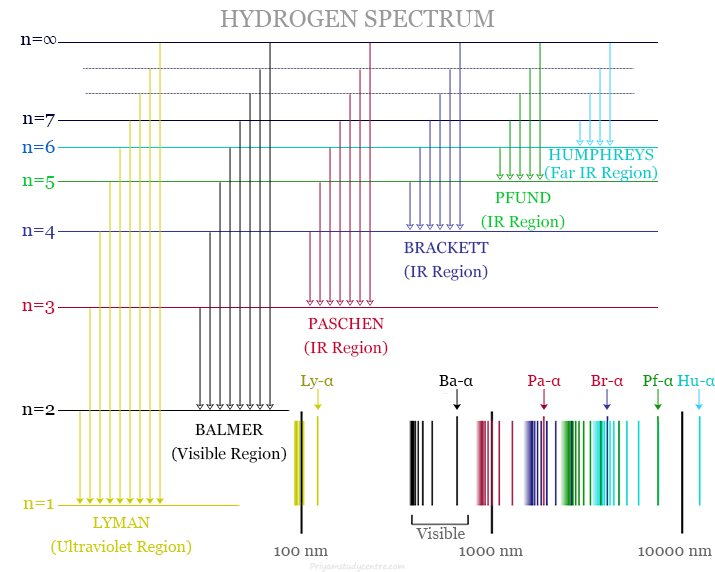
The study of the hydrogen spectrum provides early success in quantum mechanics. The atomic hydrogen emission spectrum provides various spectral lines with wavelengths given by the Rydberg formula. The observed spectral lines in the hydrogen spectrum are due to the atomic transitions between different energy levels.
Modern implementations of atomic spectroscopy are used for studying various visible and ultraviolet transitions. The modern implementation techniques of atomic spectroscopy may include:
- Flame emission spectroscopy
- Inductively coupled plasma atomic emission spectroscopy
- Glow discharge spectroscopy
- Microwave-induced plasma spectroscopy
- ARC emission spectroscopy
- X-ray and X-ray fluorescence spectroscopy
Molecular Spectroscopy
Molecular spectroscopy can be obtained by the interaction between molecules and electromagnetic radiations to generate an absorption or emission pattern of the spectrum. The mechanisms involved in molecular spectra are similar to those of atomic spectra. However, molecular spectra are more complicated than atomic spectra.
This type of spectroscopy may be used to gather information about the structure or composition of a chemical compound. Molecular spectra can be obtained due to the rotation, vibration, and electronic excitation of a molecule.
- Rotational or microwave spectra may be obtained due to the collective motions of the atomic nuclei in the microwave and millimeter-wave regions.
- Vibration motions of spectra are obtained due to relative motions of the atomic nuclei and studied in infrared and Raman spectroscopy.
- Electronic excitations can be studied in the visible and ultraviolet regions of the spectrum or fluorescence spectroscopy.
Spectroscopy Analysis
Absorption, emission, reflection, and scattering are the most common types of interaction between light and the analyzed material that are studied in various spectroscopic methods.
Types of Electromagnetic spectroscopy that can be used in the analysis of various samples are distinguished by their specific applications or implementations. The most important types of spectroscopy with their properties and applications are explained below,
Ultraviolet Visible Spectroscopy
The measurement of absorbance or transmittance of radiation in the ultraviolet-visible region of the spectrum is used for quantitative and qualitative analysis of chemical species. The absorbance of such species proceeds in two stages,
- The first stage M + hν → M∗ involves the excitation of the species by absorption of photons (hν) with a limited lifetime.
- The second stage is the relaxation that converts M∗ to a new species by a photochemical reaction.
Absorption in the ultraviolet-visible region leads to the excitation of bonding electrons. Therefore, the absorption peaks can be created with the kind of bonds existing in the species. Ultraviolet or visible spectroscopy or electronic spectroscopy is primarily used for the analysis of multiple bonds or aromatic conjugation within organic molecules.
Ultraviolet-visible spectroscopy is also used in various fields of science such as biochemistry or biological chemistry for DNA and RNA analysis, pharmaceutical analysis, bacterial culture, and beverage analysis.
Infrared (IR) spectroscopy
Infrared (IR) spectroscopy is an absorption method widely used in qualitative and quantitative analysis of electromagnetic spectrum lying in the infrared region. The infrared spectrum may alter the vibrational and rotational states of covalent bonds within the molecules.
IR spectrometer is a useful tool for the structure elucidation of organic compounds. It is also used for quantitative analysis of atmospheric pollutants such as carbon dioxide present in our environment.
The infrared spectrum is classified into three types namely, near-infrared, far-infrared, and mid-infrared.
- Near-infrared: It ranges between 12800 to 4000 cm−1
- Mid-infrared: It ranges from 4000 to 200 cm−1
- Far-infrared: It ranges from 200 to 10 cm−1
Molecules that undergo a net change in the dipole moment during vibrational and rotational motions are IR-active molecules. Therefore, homonuclear molecules such as oxygen (O2), nitrogen (N2), or chlorine (Cl2) are IR inactive because there is no net change in the dipole moment.
Molecular vibrations in IR active molecules can occur in two ways such as stretching or bending. Stretching refers to continuously changing distances in a bond between two atoms but bending refers to a change in the angle between two bonds. Bending motions of IR active molecules may include scissoring, rocking, wagging, and twisting.
The infrared spectrometers for analyzing samples operate according to very simple principles. In IR instruments, a Nernst or Glower’s lamp is used as a light source. An IR monochromator is usually a grating or a prism made of alkali halides.
A detector used for IR radiation is a thermocouple or bolometer. In IR spectroscopy instruments, the monochromator is kept before the detector and after the sample to avoid stray light.
Fourier transform infrared spectrophotometer (FTIR)
Fourier transform infrared spectroscopy (FTIR) is an analytical technique used for the analysis of an infrared spectrum of absorption or emission of a solid, liquid, or gas. It is widely used for the analysis of organic compounds, polymers mainly biopolymers, petrochemical substances, pharmaceuticals, and food items.
An FTIR instrument provides the same information as a simple spectrophotometer gives. However, the performance of FTIR is much more with respect to its speed, sensitivity, and much smaller requirements of samples.
In a Fourier transform infrared spectroscopy (FTIR) instrument, infrared radiation is split into two beams. One of these beams is kept static while the other is moving. Therefore, these two beams are then combined to get a modulated beam. Such a modulated beam provides either more light energy or less light energy for a given wavelength.
The modulated beam is passed through the sample and then Fourier-transformed by a computer to give an infrared spectrum. Modern FTIR spectrometer from the Perkin Elmer is controlled by a personal computer running Infrared Data Manager software. It also allows users to provide an analytical-grade instrument with a variable resolution of 2 cm−1 and a frequency range from 7800 to 350 cm−1.
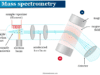
Mass Spectroscopy
Mass spectrometry is an analytical tool that uses biologists and chemists to calculate the mass-to-charge ratio of one or more molecules present in analyzing samples. Such measurements may be used to determine the precise molecular weight of the sample components in chemistry, biochemistry, pharmacy, medicine, and many related fields of science.
Mass spectrometry is not a true spectroscopic technique because the absorption of electromagnetic energy is not involved in any way. The mass spectrometer bombarded a neutral sample of volatile molecules with high-energy electrons. Therefore, electrons of energy are greater than the ionization energy for most of the compounds. It is around 8 to 13 electron volts (eV) or 185 to 300 kcal mol−1.
Mass Spectrometer
Mass spectrometry can be used to identify molecules within a sample and detect its impurities. A mass spectrometer can use an ionization source, mass analyzer, and ion detection system for their measurements.
- Ionization source: It converts molecules to gas-phase ions through vaporization before manipulating them with external electric and magnetic fields.
- Mass analyzer: It sorts and separates ions according to their mass-to-charge ratios using acceleration and deflection.
- Ion detection system: It measures ions separated by the mass analyzer and sends the data to a system where mass-to-charge ratio (m/z) ratios are stored.
In the final stage of mass spectrometry, the mass-to-charge ratios of a sample’s ions are plotted against their intensities. The produced peaks indicate the relative abundance of the sample’s molecular components.
Electron Spin Resonance (ESR) Spectroscopy
Electron spin resonance (ESR) or electron paramagnetic resonance (EPR) spectroscopy is a method used for studying paramagnetic substances that exhibit characteristic magnetic properties. The electron spin resonance phenomenon may be shown by atoms having an odd electron, ions having partially filled inner energy levels, and molecules that carry angular momentum.
The basic principle, methodology, and instrumentation of electron spin resonance spectroscopy are more or less similar to that of nuclear magnetic resonance spectroscopy. The only exception is the absence of chemical shift phenomena in ESR spectroscopy.
Electron spin resonance (ESR) spectroscopy has been uses widely for analysis of free radicals, even at very low concentration. It is also useful in the determination of trace amounts of paramagnetic ions in biological samples. Similarly, odd electron molecules, transition metal complexes, lanthanide ions, and triple-state molecules have been studied by this technique.
Electron spin resonance (ESR) spectroscopy is used for the analysis of stable molecules containing odd electrons and having the capacity to react readily with amino acids. ESR of such compounds provides valuable information about the structure, polarity, viscosity, phase transformation, and chemical reactivity.
Raman Spectroscopy
Raman spectroscopy is an analytical tool where scattered light is used to measure the vibrational energy modes of an analyzed sample. In 1928, Sir C.V. Raman discovered that when a beam of monochromatic light passes through a substance, the scattered light contains some additional frequencies over and above that of the incident frequency. It is known as the Ramman effect which occurs when a light beam is deflected by molecules.
Raman spectroscopy is related to the vibrational or rotational transition but in a different manner. In Raman spectroscopy, only the scattering is measured but it does not measure the absorption of radiation. When the incoming radiant energy strikes upon particles that are suspended in a medium having a refractive index different from that of the suspended particles, the light transmitted at an angle other than 180° from the incident light is said to be scattered.
A highly monochromatic light source is essential to get small Raman shift. Therefore, Raman spectroscopy measurement is usually carried out in the visible region where high dispersion is easily available with less expensive instruments.
Raman spectroscopy is a very useful tool that provides information about chemical structure, phase and polymorphy, crystallinity, and molecular interactions. Therefore, the size shape, and concentration of colloid particles and suspension can be measured by Raman scattering.
Applications of Spectroscopy
Spectroscopy can be used in various fields of science such as medicine, physics, chemistry, and astronomy. The main applications of Electromagnetic spectroscopy in such fields of science may include:
- Spectroscopy is used for studying the structure of atoms and molecules.
- Electromagnetic spectroscopy is an analytical tool used for studying and finding the constituents in a material having an unknown chemical composition. The emission spectrum has been used to analyze the concentration of a trace element present in a material.
- In chemistry and medicinal chemistry, the spectroscopic technique can be used to identify the chemical composition of studying materials and drugs.
- In biochemistry, spectroscopic methods are used for the characterization of proteins.
- In astronomy, the spectral emission lines of distant galaxies can be used to study and discover the universe that expanding rapidly.
- It is used for determining the metabolic structure of a muscle.
- In environmental chemistry, spectroscopy techniques are used in various environmental analyses. For example, atomic emission and absorption spectroscopy in the visible and ultraviolet regions can be used to determine metals in water or solids samples.
- Infrared spectroscopy also plays a significant role in environmental analysis. Therefore, long-range infrared sensors can be used to determine the concentration of certain toxic compounds in the air.
- In electromagnetic spectroscopy, x-ray methods have been used to determine the atomic composition of solid materials. It determines metal concentrations in particulate matter from the air and soil samples.
- The spectroscopy technique in biological chemistry and environmental science may also be used to monitor the dissolved oxygen content in freshwater and marine ecosystems.
Frequently Asked Questions
What is spectroscopy?
Spectroscopy is the study of the spectrum produced by matter when interacting or emitting electromagnetic radiation. Scientists commonly use spectrometer instruments for the separation and measurement of spectral components of any substance through the analysis of its light properties. We commonly use atomic absorption spectroscopy (AAS), atomic emission spectroscopy (AES), and mass spectrometry for the analysis of various chemical samples.
How mass spectroscopy works?
Mass spectroscopy is not a true spectroscopic technique because the absorption of electromagnetic energy is not involved in any way. Mass spectroscopy works by bombarding a neutral sample of volatile molecules with high-energy electrons. Therefore, electrons of energy are greater than the ionization energy for most of the compounds. It is around 8 to 13 electron volts (eV) or 185 to 300 kcal mol−1.
What is Raman spectroscopy?
Raman spectroscopy is an analytical technique where scattered light can change the frequency due to the interaction between the incident photons and the vibrational energy levels of a sample.
Raman spectroscopy is a very useful tool that provides information about chemical structure, phase and polymorphy, crystallinity, and molecular interactions. Therefore, the size shape, and concentration of colloid particles and suspension can be measured by Raman scattering.
What is Fourier transform infrared spectroscopy?
Fourier transform infrared spectroscopy (FTIR) is an analytical technique used for the analysis of an infrared spectrum of absorption or emission of a solid, liquid, or gas. It is widely used for the analysis of organic compounds, polymers mainly biopolymers, petrochemical substances, pharmaceuticals, and food items.
An FTIR instrument provides the same information as a simple spectrophotometer gives. However, the performance of FTIR is much more with respect to its speed, sensitivity, and much smaller requirements of samples.