Isopropyl Alcohol (2 Propanol)
Propanol (propyl alcohol and isopropanol) is used widely for the production of various organic compounds, intermediates, and solvents in the chemical and pharmaceutical industries. It has two isomers, n-propanol or n-propyl alcohol or 1-propanol and isopropanol or 2-propanol with the chemical formula C3H8O or C3H7OH. Propanol has a low toxicity for human health but high concentration may irritate the eyes, nose, and throat. Both the isomers are less toxic than methanol and ethanol. Propanol is an alcohol with a three-carbon chain and an alcoholic group in its structure. Propanol is used widely in antiseptics, cleaning agents, astringents, and muscle ache treatments. Currently, propanol is used for the production of propylene which is an important substance in chemical industries.
Uses of Propanol
Propanol has two isomers, 1−propanol or n−propyl alcohol and 2−propanol or isopropanol. Both these organic compounds are widely used as solvents and intermediates in the pharmaceutical and chemical industries. They are also used widely in electronic cleaning products.
Isopropanol (one of these two isomers) is used to manufacture various types of organic compounds such as acetone, glycerol, isopropyl acetate, etc.
The most common uses of propanol (n-propyl alcohol and isopropanol) are
- Solvents/intermediate
- Medicinal use
- Motor fuel
Solvents
Propanol is used as a solvent in many pharmaceutical and chemical processes.
- Two isomeric compounds, n−propyl alcohol, and isopropanol can dissolve a wide range of non−polar chemical compounds. Therefore, they can be used to dissolve ethyl cellulose, polyvinyl butyral, essential and other oils, alkaloids, gums, resins, etc.
- It is also used for the production of other solvents such as antifreeze, lacquer, soap solutions, dye solutions, etc.
- Isopropanol is a green solvent. The isopropanol solution is used for the synthesis of amines and their derivatives by hydrogenation of nitro compounds. These amines and their derivatives are used for the synthesis of different pharmaceuticals, polymers, and agrochemical products.
Medicinal Use
In the medicine or healthcare industry, it is used widely for the production of rubbing alcohol, disinfectants, cleaning products, hand sanitizers, etc.
- Generally, we used 60–90 percent solutions of isopropanol and water for the production of hand sanitizer.
- We also used a topical rub of propanol to relieve muscle pain.
- Early days, it can be used in anesthesia but it causes several types of toxic side effects on humans. Therefore, propanol is not safe to use in anesthesia.
Biofuel
Propanol has a high octane number and is quite suitable for use as a biofuel in engines. It is also less toxic and less volatile compared to methanol. However, the use of propanol as a biofuel does not gain much more attention due to its production cost compared to gasoline.
The energy density of propyl alcohol is not much higher than ethanol and not much lower than butanol. Therefore, commonly we use a mixture of propanol–butanol–ethanol as a fuel for engines.
Production Process
Both the compounds, n-propyl alcohol, and isopropanol are industrially attractive chemical compounds that can be produced by many chemical processes.
n-Propyl Alcohol (1−Propanol)
n-Propyl alcohol is originally obtained from fusel oil but it is now produced by the hydrogenation of carbon dioxide.
A more recent method for the production of n-propyl alcohol is the catalytic reduction of propargyl alcohol obtained from acetylene and formaldehyde.
CH≡CCH2OH + 2H2 → 2 CH3CH2CH2OH
The largest use of n-propyl alcohol as a solvent in cosmetics, pharmaceuticals, and lacquer industries.
Isopropyl Alcohol (2−Propanol)
Isopropyl alcohol can be prepared industrially by the following process,
- Isopropyl alcohol (2−propanol) can be prepared industrially by passing propane from cracked petroleum into concentrated sulfuric acid.
2 CH3CH=CH2 + H2SO4 → [(CH3)2CHO]2SO2 - The product obtained from the above reaction can be diluted with water and distilled off to form isopropanol.
[(CH3)2CHO]2SO2 + 2 H2O → 2 (CH3)2CHOH - The by-product isopropyl ether is also obtained by this production process.
Isopropanol is also prepared by direct hydration by passing a mixture of propene and steam at 220−220 °C and under pressure (220 °C) over a catalyst of tungsten oxide plus zinc oxide on a silica carrier. Commercially, isopropanol is a good chemical reagent for the production of acetone, ketene, and high-octane fuel.
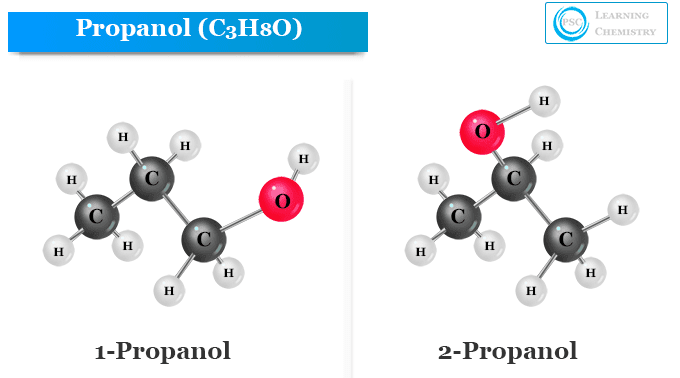
Structure of Propanol
Propanol is a monohydric alcohol that contains two isomeric forms. Monohydric alcohols are organic compounds that contain one hydroxyl group (−OH) that is attached to the carbon atom in an alkyl group or hydrocarbon chain.
Propanol is alcohol with the general molecular formula C3H8O or C3H7OH. It has two isomeric forms n-propyl alcohol and isopropanol with two different structures.
Both forms contain the same carbon skeleton (carbon chain) and functional groups in their structure. The position of the hydroxyl group is different places for n-propyl alcohol and isopropanol. Therefore, position isomerism can be seen in n-propyl alcohol and isopropanol.
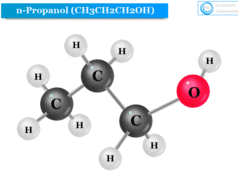
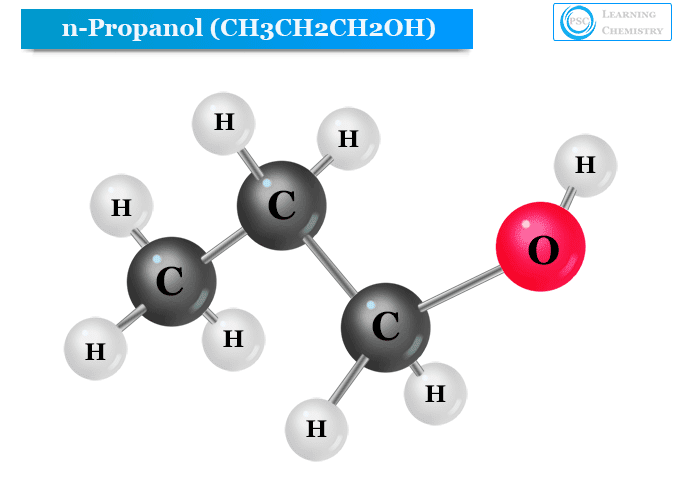
Structure of n-Propyl Alcohol
The hydroxyl group is directly attached to the primary carbon atom of n-propyl alcohol. Therefore, n-propyl alcohol (1−propanol) is a primary alcohol that has the chemical formula CH3CH2CH2OH or PrOH or n-PrOH.
The structure of n-propyl alcohol is given below the picture,
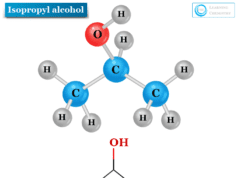
Structure of Isopropanol
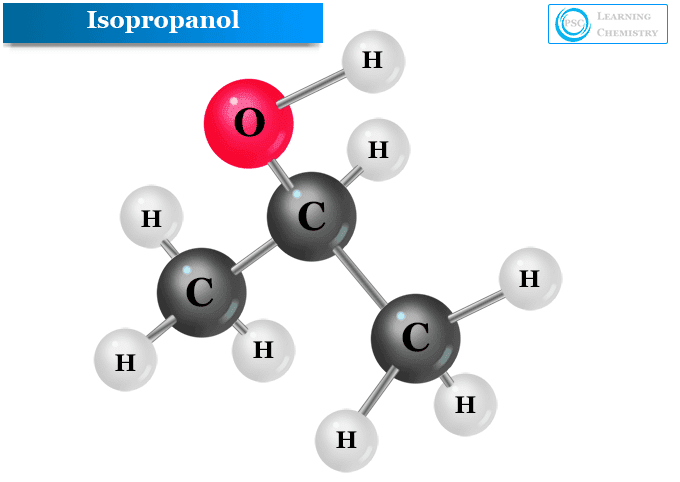
The chemical formula of isopropanol is written as CH3CHOHCH3 or (CH3)2CHOH. Hence an isopropyl group is directly attached to the hydroxyl group in the isopropanol or isopropyl alcohol molecule.
The structure of isopropanol or 2-propanol molecule is given below the picture,
Properties of Propanol
Two isomeric forms of propanol (n-propyl alcohol and isopropanol) are miscible in water, ethanol, and ether. Some common properties of these two alcohols (n−propyl alcohol and isopropyl alcohol) are given below in the table,
| Properties | n−propyl alcohol | Isopropanol |
| Molecular Formula | CH3CH2CH2OH | (CH3)2CHOH |
| Molecular Weight | 60.096 g/mol | |
| Density | 0.803 g/cm3 | 0.786 g/cm3 |
| Boiling Point | 97 to 98 °C | 82.6 °C |
| Melting Point | −126 °C | −89 °C |
| Appearance | Colorless liquid | |
| Solubility in water | Miscible in water | |
| CAS Number | 71-23-8 | 67-63-0 |
| Dipole moment | 1.68 D | |
Chemical Reactions
Propanol reacts with organic and inorganic acids to form esters.
CH3COOH + CH3CH2CH2OH → CH3COOCH2CH2CH3
Strongly electropositive metals (potassium, sodium, magnesium, aluminum, zinc) liberate hydrogen from alcohol to form alkoxides. For example, sodium reacts with propanol to form sodium propoxide.
2 C3H7OH + 2 Na → 2 C2H5O−Na+ + H2
n−propyl alcohol and isopropanol may be acetylated with acetyl chloride. For example, n−propyl alcohol gives propyl acetate.
CH3COCl + C3H7OH → CH3CO2C2H5 + HCl
Propyl alcohol may be oxidized and the products of oxidation depend on the class of the alcohol and the nature of the oxidizing agent.
Isopropyl alcohol can be dehydrated by concentrated sulfuric acid at 170 °C to form propane.
(CH3)2CHOH → CH2=CHCH3
Oxidation of n-propyl alcohol by Na2Cr2O7 and H2SO4 gives propionaldehyde while isopropyl alcohol can be oxidized by chromic acid to form acetone.
C3H7OH + [O] → C3H7CHO
(CH3)2CHOH → CH3COCH3
Health Hazards
Both n−propyl alcohol and isopropanol are low toxicity for human health but high concentrations may irritate the eyes, nose, and throat.
- If we inhale, propyl alcohol could be dangerous for human health. It can seriously damage your eyes, impact the central nervous system, and irritate the respiratory tract.
- n-Propyl alcohol poisoning can seriously damage your central nervous system (CNS). The most common symptoms of n-propyl alcohol poisoning are confusion, reduced consciousness, slowed pulse and breathing, etc. Therefore, it can be used in hand sanitizer.
Frequently Asked Questions (FAQs)
What are the uses of propanol?
As a solvent, two isomeric forms of propanol are used widely in pharmaceuticals, floor polishes, dental lotions, lacquers, printing inks, natural gums, pigments, intermediates, dye solutions, antifreeze, gasoline additives, paint additives, etc.
- n-Propanol is used commonly for making propanoic acid, toilet lotions, soaps, dye solutions, window cleaners, etc.
- Isopropyl alcohol is used commonly for making esters, acetone, keten, printing ink, etc.
What is the real name of propanol?
Propanol has two isomeric forms of alcohol and the common names of these two forms are n-propyl alcohol and isopyl alcohol.
- The IUPAC name of n-propyl alcohol is propan-1-ol or 1-propanol.
- Similarly, the IUPAC name of propanol is propan-2-ol or 2-propanol.
How to convert propene to propanol?
Propene can be converted to propanol when passed over heated alumina. It may also converted by concentrated sulfuric acid at about 170 C.
Isomerization usually occurs when dehydrated by sulfuric acid or alumina. However, in dehydration by alumina, isomerization is suppressed by the addition of a small amount of pyridine.
What is the formula of propanol?
The general molecular formula of propanol is C3H8O. However, it has two isomeric forms. n-Propyl alcohol is one of two isomeric alcohols with the chemical formula CH3CH2CH2OH. Isopropyl alcohol is the second isomer with the chemical formula (CH3)2CHOH.
How to distinguish 1-propanol and 2-propanol?
Propanol has two isomeric forms 1-propanol and 2-propanol two different structures. Both forms contain the same carbon skeleton (carbon chain) and functional groups in their structure. The position of the hydroxyl group is different places for these two alcohols. Therefore, position isomerism can be seen in 1-propanol and 2-propanol.
n-Propyl alcohol is a primary alcohol but isopropyl alcohol is a secondary alcohol. Therefore, they can be distinguished chemically by oxidation.
- On oxidation, n-propyl alcohol gives propanaldehyde and on further oxidation, it gives an acid. Both aldehyde and acid obtained during oxidation contain the same number of carbon atoms as the original alcohol.
- On oxidation, isopropanol gives acetone. Acetone can be difficult to oxidize but further oxidation by suitable reagents gives a mixture of acids (formic acid and acetic acid) where each contains fewer carbon atoms than the original alcohol or 2-propanol.