Aluminum Metal
Aluminum (symbol Al) or aluminium is a chemical element or silvery-white, lustrous, low melting, soft metal of Group 13 (IIIA) or boron family of the periodic table uses widely in our everyday life. The metal aluminum is the third most abundant chemical element after oxygen and silicon. The metal aluminum differs from the first member of Group 13 (boron) due to its chemical properties, high reactivity, and cationic chemistry in an aqueous solution. Aluminium is the most abundant metallic element in the earth’s crust (8 percent by weight). It combines most of the nonmetallic elements like nitrogen, sulfur, and halogens. It forms intermetallic aluminum compounds with a large number of metals.
Properties of Aluminum
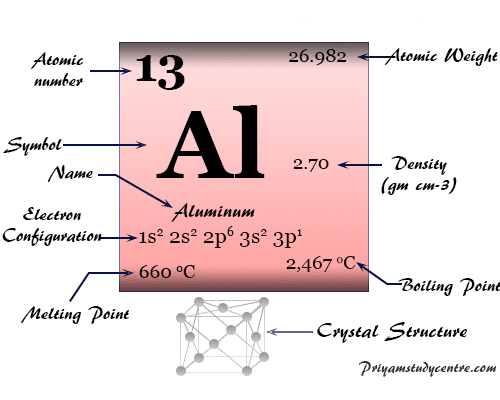
Face-centered cubic crystal lattice (fcc), aluminum has the chemical symbol Al, atomic number 13. The name aluminum comes from the Latin word alumen, which describes the aluminum potassium sulfate, KAl(SO4)2, 12H2O.
| Aluminum | |||
| Symbol | Al | ||
| Discovery | Hans Oersted in 1825 | ||
| Name derived from | The Latin word for alum alumen means bitter salt | ||
| Common isotope | 13Al27 | ||
| Oxidation states | −2, −1, +1, +2, +3 | ||
| CAS number | 7429-90-5 | ||
| Periodic properties | |||
| Atomic number | 13 | ||
| Relative atomic mass | 26.982 | ||
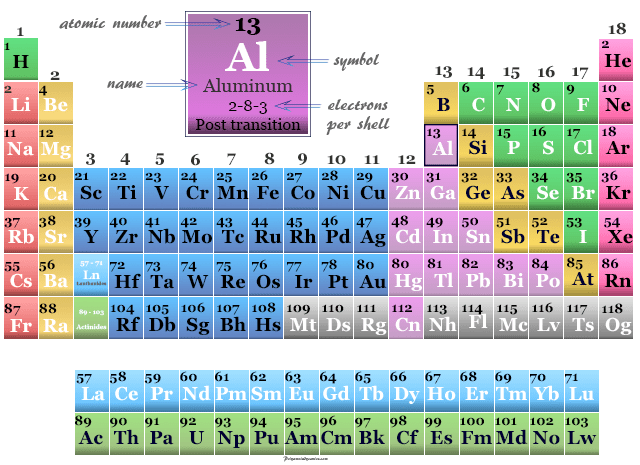
| Electron per cell | 2, 8, 3 | ||
| Electronic Configuration | [Ne] 3s2 3p1 | ||
| Block | p-block | ||
| Group | 13 | ||
| Period | 3 | ||
| Physical properties | |||
| State at 20 °C | Solid | ||
| Melting point | 660.3 °C, 1220.6 °F, 933.5 K | ||
| Boiling point | 2519 °C, 4566 °F, 2792 K | ||
| Molar heat capacity | 16.443 J mol−1 K−1 | ||
| Crystal structure | face-centered cubic (fcc) | ||
| Density | 2.70 g/cm3 | ||
| Electrical resistivity | 26.5 nΩ m | ||
| Atomic properties | |||
| Atomic radius (non-bonded) | 1.84 Å | ||
| Covalent radius | 1.24 Å | ||
| Electronegativity | 1.61 (Pauling scale) | ||
| Electron affinity | 41.762 kJ mol−1 | ||
| Ionization energy (kJ/mol) | 1st | 2nd | 3rd |
| 577.54 | 1816.68 | 2744.78 | |
The common oxidation number or states of metal +1 +2 and +3. Due to decreases in lattice energy with increasing size, aluminium should be likely to form an Al+3 ion. Due to large ionization energy not compensated by weak lattice energy, it generally attains a +3 state by covalent bonding.
Aluminum in the Periodic Table
Aluminum is found in group 13 and period 3 of the periodic table. It is a metalloid that lies between boron and gallium.
Where is Aluminum Found?
The most abundant element, aluminum found in various silicate minerals like mica, feldspar, kaolinite, and clay. The only fluoride mineral is cryolite (Na3AlF6), found in Greenland.
The most important minerals found in nature as oxide and anhydrous hydrated oxides. Oxide like Corundum (Al2O3) is used in Gems (ruby, sapphire) due to its hardness.
Where is Bauxite Found?
The chief commercial source of metallic aluminum is hydroxo oxide like Al2O3, xH2O, or bauxite. A workable bauxite sample contains 40 to 60 percent metal aluminum oxide (Al2O3), 12 to 30 percent water, 1 to 15 percent iron oxide (Fe2O3), and 3 to 4 percent titanium oxide (TiO2).
Rich deposits of bauxite are found in Australia, Brazil, Guinea, Jamaica, France, the United States, Russia, India, and several African countries.
Who Discovered Aluminum?
Alum was the compound of aluminum widely used in Rome for the production of different types of medicine and dyes in textiles.
British chemist Sir Humphry Davy failed to isolate the metal but suggested the name of the element aluminum or alumium.
In 1825 Denmark physicist Hans Christian Orsted first discovered the metal by reducing aluminum chloride (AlCl3) with potassium amalgam. Commercial production of the element started in 1954 by electrolysis of NaAlCl4.
Aluminum Production Process
It can be produced electrolytically from bauxite in electrochemical cells.
Purified bauxite (melting point 2050 °C) is dissolved in cryolite and electrolyzed at 950 °C in carbon lined steel cells.
A mixture of cryolite, CaF2, AlF3, Al2O3, and Li2CO3 in the electrolyte is used because it has a lower melting point, permits larger current flow, and reduces fluorine emission.
The electrode reaction for the production of aluminum,
Cathode: 2 Al+3 + 6e → 2 Al
Anode: 3 O−2 → 3 O + 6e
The molten aluminum sinks to the bottom of the cell and drains out. The surface of the electrolyte was covered with a layer of coke, which oxidized evolving oxygen for the protection of valuable electrodes.
Chemical Properties
It is not attacked by water owing to the presence of a protective coating. If the film is removed by amalgamation, water forms [Al(OH)3 + H2]. All the elements of Group 13 have different chemical properties and reactivity.
- For example, Al and Ga dissolve in aqueous hydrochloric acid giving M+3 ions but Tl does not due to the instability of TlCl.
- Al is scarcely attacked by dilute sulfuric acid but concentrated acid is reduced to sulfur dioxide.
- Dilute and concentrated nitric acid render aluminum passive by forming a protective oxide layer. Al dissolves in hot aqueous alkali (lithium, sodium, and potassium) to form [Al(OH)4]− and hydrogen.
Chemical Compounds
The electronic structure of Al suggests that it forms mostly trivalent chemical compounds. The aluminum compounds in the +3 state are dominantly by covalent chemical bonding. In suitable conditions, they formed +2 and +1 compounds like AlCl, Al2O, and AlO.
In learning chemistry, the tripositive ion (Al+3) ions may be stabilized in aqueous solutions through strong hydration energy, usually, six water molecules held strongly by the Al+3 ion.
Aluminum Hydride
The hydride of Al is a colorless involatile solid, thermally unstable above 150-200 °C. It is a strong reducing agent and adducts with strong Lewis bases.
The tetrahydro complex of Al like LiAlH4 is an equally important chemical compound and widely uses in organic chemistry or the reduction of hydrocarbon.
Aluminum Halides
All four trihalides of aluminum have the general formula AlX3, where X = fluorine, chlorine, bromine, and iodine. All the compounds may be formed during a direct combination of the metal with halides.
The properties of the trihalides are very wide. For example, fluoride is an ionic crystalline solid.
What is Alumina?
Alumina is an important chemical compound of aluminum with two structural forms, α-Al2O3 and ϒ-Al2O3. The α-Al2O3 occurs in mostly nature as corundum which is resistant to water and acids and is used in Gems.
The γ-alumina is distorted, less dense, and soft with high surface tension in the liquid form. These qualities of γ-alumina are excellent reagents for dehydration, decolorization, and absorption chronograph.
β-alumina is now being developed as a ceramic ion conductor useful for high-energy batteries. Therefore, large quantities of alumina are used for the commercial production of aluminum.
Uses of Aluminum
In recent years aluminum acquired a high position among the metals due to its several exclusive properties,
- it is easily malleable and non-toxic
- fairly corrosion resistant in dry conditions
- good thermal and electrical conductors.
- it is cheap in price
The corrosion-resistant properties are due to the protective oxide layer in metal. Pure aluminum is quite soft and weak but the commercial form contains small amounts of silicon and carbon is hard and strong.
The mechanical strength of aluminium significantly increases by alloying with metals like iron, copper, manganese, silicon, magnesium, or zinc. Aluminum alloys are used mostly in construction work, transportation, containers and packaging, electrical power lines, machinery, and the paint industry.
- The metal and its alloys are now extensively used in aircraft, rocket fuels, trucks, trailers, and car frames.
- In making windows, doors, and building panels we also used it.
- In making machinery, utensils, and other household products including furniture also made of aluminum alloy.