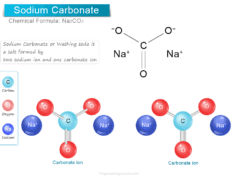
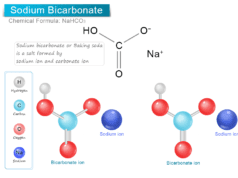
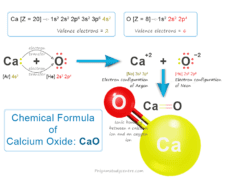
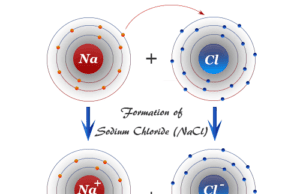
Glycerol (Glycerine)
Glycerol, also called glycerine or glycerin is a naturally occurring organic trihydric alcohol that has a variety of benefits and uses in the production of skin hydration and care products, antifreeze agents, food and medicine, and polymers. Organic glycerol obtained from vegetable oils and animal sources is a colorless, odorless, viscous liquid with high viscosity, sweet-tasting solution with the molecular formula HOCH2CHOHCH2OH. It is soluble in water and non-toxic in low concentrations.
The alcohol glycerol is a sweet testing viscous liquid obtained generally from plants and animal sources. It occurs in plants and animals as triglycerides, esters of glycerol with long-chain carboxylic acids.
Glycerine is an impotent chemical solution that is used widely in the food industry as a solvent or sweet testing liquid, pharmaceuticals, and personal and skin care products. It is a basic ingredient in producing gums, resins, and exterior house paints. Glycerin reacted with nitric acid and sulfuric acid to form explosive nitroglycerin.
What is Vegetable Glycerin?
Vegetable glycerin or glycerol is a clear, odorless, fatty liquid obtained from vegetable fats. It is a variant that is generally made from plant oils.
It was accidentally discovered more than two centuries ago, when Scheele, a Swedish chemist heated a mixture of olive oil and lead monoxide (PbO) in a container. Vegetable glycerin can be made by heating triglyceride-rich vegetable fats (palm, soy, and coconut oils) under pressure or with strong alkalis such as sodium hydroxide or potassium hydroxide.
Along with other uses, vegetable glycerin is particularly popular in the cosmetic industry such as such as skin care, hair care, and oral hygiene products. Vegetable glycerin is generally safe and non-toxic but if consumed orally, it may have some side effects.
Structure of Glycerol Molecule
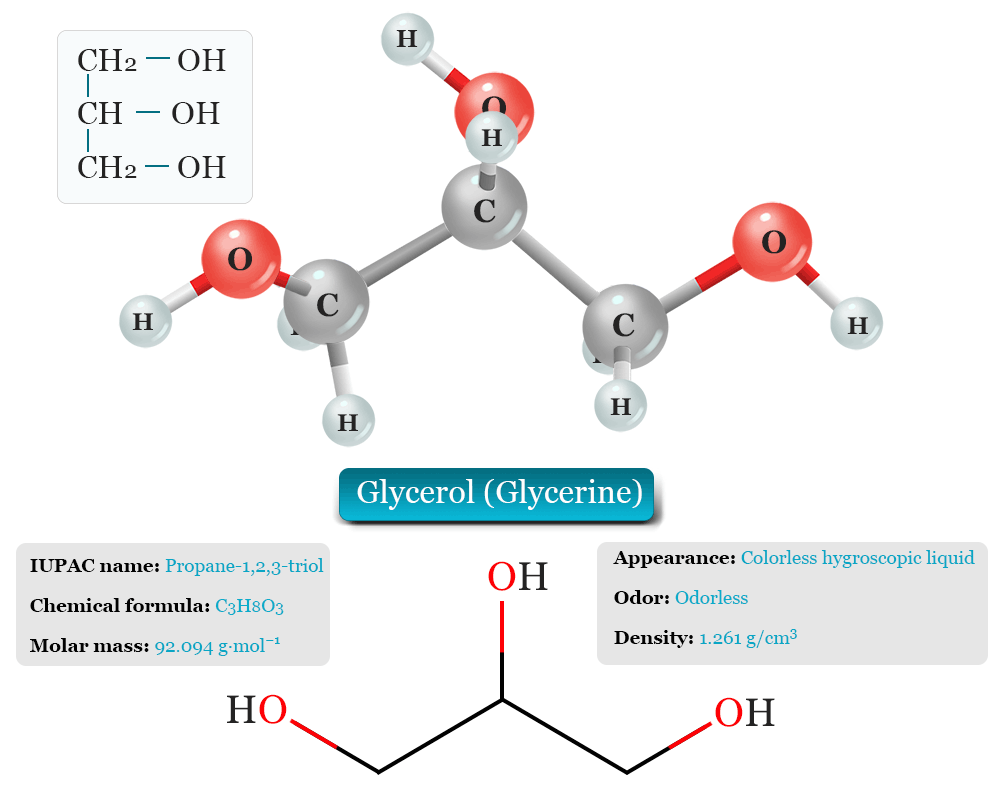
Glycerol, also called glycerine or glycerin is a naturally occurring trihydric alcohol that uses three carbon atoms, eight hydrogen atoms, and three oxygen atoms to constitute its structure. It has three carbon chains with 3 hydroxyl groups (OH) attached to each. The carbon atoms in the glycerine molecule are sp3 hybridized.
It has a molecular weight of 92.094 g/mol and chemical formula C3H8O3. The IUPAC name of glycerol is 1, 2, 3-Propanetriol or 1, 2, 3-Trihydroxypropane.
Properties of Glycerol
Glycerol is a colorless, odorless, syrupy liquid with a sweet taste. Glycerol is a hygroscopic liquid that uses to absorb moisture from the air. It is miscible with water and alcohol (methanol or ethanol), but insoluble in chloroform and ether.
Physical Properties
Glycerin is the only important trihydric alcohol that is soluble in water and hygroscopic in nature. It is a highly viscous liquid that absorbs water from the air. Glycerin is a type of carbohydrate that is classified as a sugar alcohol or polyol.
| IUPAC Name | Propane-1,2,3-triol |
| CAS Number | 56-81-5 |
| Chemical formula | C3H8O3 |
| Molar mass | 92.094 g·mol−1 |
| Appearance | Colorless hygroscopic liquid |
| Solubility | Soluble in water |
| Density | 1.261 g/cm3 |
| Melting point | 17.8 °C |
| Boiling point | 290 °C |
| Viscosity | 1.412 Pa·s at 20 °C |
Chemical Properties
Glycerol contains one secondary and two primary alcoholic groups in its structure. Therefore, it undergoes many of the reactions to be expected of these types of alcohols.
- When glycerol solution is treated with sodium, one primary alcoholic group is readily attacked, and the other primary alcoholic group is less readily attacked. However, the secondary alcoholic group is not attacked at all.
- Both α- and β-glycerol monochlorohydrin are formed when passing hydrogen chloride into glycerol at 110 °C.
- When glycerol is treated with monocarboxylic acids, mono-, di, or tri-esters are obtained according to the amount of acids we use.
- Nitroglycerine is manufactured by adding glycerol solution in a thin stem to a cold mixture of concentrated nitric and sulfuric acid. It is an ester but not a nitro compound.
- When glycerine is heated with formic acid or oxalic acid at 260 °C, it is converted into allyl alcohol. The polymer glyptal is formed when glycerine is heated with phthalic anhydride.
- When boric acid is added to an aqueous glycerine solution, a complex is produced which has higher electrical conductivity than boric acid itself.
- When heated with potassium hydrogen sulfate, it dehydrates to form acraldehyde.
Production of Glycerol
We can obtain pure glycerol from vegetable or animal-based sources. Glycerol was first made in 1779 by K. W. Scheele, a Swedish chemist when he was boiling a mixture of olive oil and another chemical containing lead such as PbO in a container.
It is the only important trihydric alcohol that occurs in almost all animal and vegetable oils and fats. It occurs as triglycerides and glyceryl esters of mainly palmitic, stearic, and oleic acids.
From Natural Sources
Generally, a large quantity of glycerine may be obtained as a byproduct in the manufacture of soap, and it is still a commercial source of glycerol.
Nowadays, a large quantity of glycerin is produced from the hydrolysis of fats and oils. Industrially, glycerols and some fatty acids are prepared by the hydrolysis of oils and fats with water under pressure at 220 °C. The glycerol is recovered from the aqueous solution, and free fatty acids are used for the manufacture of candles.
If an oil that contains triglycerides can be saponified with sodium hydroxide or potassium hydroxide, it gives glycerol solution and sodium salt of fatty acid or soap.
From Artificial Sources
Glycerin is obtained artificially from propene in various ways. Epichlorohydrin is the most important precursor that uses for the production of glycerol. Glycerol solution can be obtained from propylene in the following way:
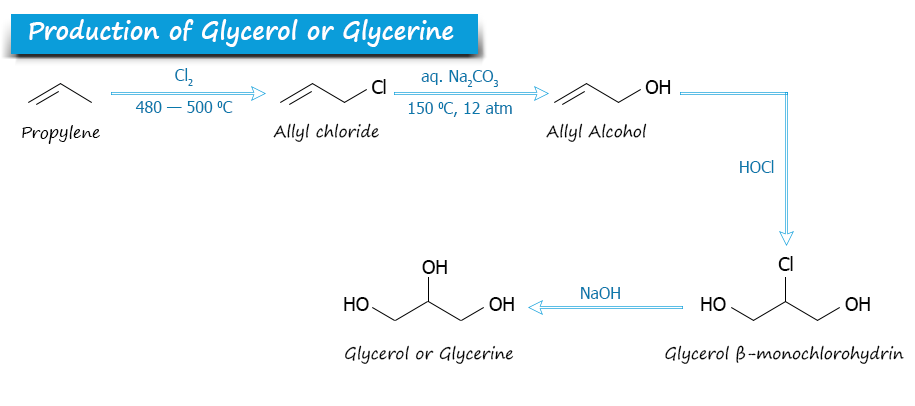
- The chlorination of propylene gives allyl chloride.
- It oxidizes with hypochlorite to produce dichlorohydrins.
- When dichlorohydrins react with a strong base, they form epichlorohydrin.
- Epichlorohydrin is then hydrolyzed to form glycerol by this process.
Glycerol can be produced synthetically from acrolein and propylene oxide. It is obtained in large quantities as a by-product in the manufacture of soap. It is still a commercial source of glycerol and is produced synthetically in the following way:

Liquid Glycerol Benefits
Glycerol obtained from vegetable oils and animal sources is beneficial for us particularly care your skin. The most common benefits of liquid glycerol may include:
- May moisturize your skin: Vegetable glycerol is particularly popular in the cosmetic industry such as skin care treatment because of its moisturizing power.
- May promote your skin health: Glycerine may improve skin health by reducing skin irritation, care against infection, and promoting wound healing.
- May reduce constipation: It can draw water into your gut. Therefore, glycerol may help digested food move through your gut more smoothly.
- May boost hydration and athletic performance: It can boost hydration and improve your athletic performance.
Uses of Glycerol
Glycerol, also called glycerin or glycerine is an important organic compound that is widely used in the food, pharmaceutical, and cosmetic industries to care for your skin.
According to a 2019 FDA report, glycerine is the third most common ingredient in personal care products after water and fragrance. The most common items where glycerin is used widely may include:
- Lipsticks
- Glycerine soaps
- Detergents
- Hair coloring products and Hair conditioners
- Skincare products ( lotions and makeup items)
- Tanning products
- Baby products
- Eye creams
- Shaving products and deodorants
- Oral care products and Oral medicines
- The sweetener in candies and cookies
- Solvent and preservative in food products
- Ice cream and toothpaste base
Glycerol in the Food Industry
In the food and beverages industries, glycerol is a humectant that reduces the loss of moisture. It is also used as a solvent and sweetener that may help to preserve your foods.
Glycerol or glycerin is a binding agent that is used for the preparation of low-fat foods such as cookies. It is also used as a thickening agent in beer, honey, vinegar, wine, and wine vinegar. It is used for the preservation of certain types of plant leaves.
Pharmaceutical and Medical Industries
Along with skin care, glycerol may also uses in medical and pharmaceutical industries to improve smoothness and lubricate medical or pharmaceutical products. It can be found in cough syrups, allergen immunotherapies, and elixirs. It is also used as a tablet hiding agent in the production of tablets.
Glycerine has moderate antimicrobial and antiviral activity. Therefore, it can be used to treat wounds and certain types of burns.
It can also used for the treatment of your eyes because when glycerine is taken orally, it can rapidly and temporarily decrease your eye pressure. In the blood bank, glycerol is used for the preservation of red blood cells before freezing.
Glycerol in Personal Care Products
Glycerol uses as a humectant that can reduce the loss of moisture in the preparation of personal care products. Glycerin can enhance the skin’s ability to absorb moisture or water from the outer environment.
Glycerin may improve the smoothness and lubrication of personal care items. Therefore, glycerine is found in various personal care items such as toothpaste, mouthwashes, skin care products, shaving cream, hair care products, soaps, and water-based personal lubricants. It is the main component in the production of glycerine soaps.
Other Uses of Glycerol
- Glycerin is used as a replacement for water in ultrasonic testing due to its good acoustic properties.
- It is a naturally occurring trihydroxy sugar alcohol that is beneficial for carbohydrate and lipid metabolism.
- Glycerine is an important component for making explosives of dynamite.
- In film industries, glycerine is a decorating agent in film scenes to avoid quick drying of wet areas.
- Like ethylene glycol, glycerol also forms strong hydrogen bonding with water. Such a type of interaction may reduce the formation of ice. Therefore, it may also used as an antifreezing agent.
- Glycerin, along with propylene glycol, is a common component for making electronic cigarettes.
- Glycerin is a basic component in the gums and resins that is used to produce many modern protective layers such as automotive enamels and exterior house paints.
Glycerol Polymer
Glycerol polymers are biodegradable and biocompatible polymers produced from glycerol through fermentation, chemical conversion, and direct polymerization. It is the major by-product obtained in biodiesel production.
Glycerine-based polymers exhibit excellent biocompatibility and uses in the following fields:
- Glycerol-based polymers such as polyglycerol ether, polyglycerol carbonates, and dendritic hyperbranched have been extensively used in pharmaceutical applications such as drug delivery, tissue implants, and as an anti-bacterial agent. However, the impurities in crude glycerol solution make it incompatible for such types of uses.
- Such types of biopolymers are crucial to avoid environmental burdens.
- It is an important additive for the polymer industry.
- It is also useful in increasing bio-based polymers’ mechanical and thermal properties.
Frequently Asked Questions (FAQ)
Glycerol is which type of alcohol?
Glycerol, also called glycerine or glycerin is a naturally occurring trihydric alcohol that contains three different types of hydroxyls or alcoholic groups in its structure.
Is glycerol a sugar?
Glycerin or glycerol is a type of carbohydrate called a sugar alcohol, or polyol. It contains the three-carbon backbone of a triglyceride. Therefore, glycerine is formed naturally through the alcoholic fermentation of sugar molecules.
Most of the glycerin we consume is produced from the hydrolysis of fats and vegetable oils. Lesser quantities are obtained through the fermentation of yeast, sugar, or starch.
Is glycerine good for your skin?
Glycerin is very useful for skin care because it acts as a humectant that allows your skin to retain moisture. After applying glycerine to the skin, it may increase skin hydration, relieve dryness, and refresh the skin’s surface.
It can also act as an emollient that helps to make your skin soft and smooth. Additionally, liquid glycerine or glycerol uses as a skin care to reduce wrinkles by attracting moisture to the top layer of the skin.