Gadolinium Element
Gadolinium is a chemical element or rare earth metal in the periodic table with the symbol Gd and atomic number 64. It is the reactive and softest lanthanide that is stored under an inert fluid to protect it from atmospheric oxygen or moisture. The f-block element gadolinium is useful to improve the workability of iron and chromium alloys. In medicinal chemistry, gadolinium compounds are used as MRI contrast agents to enhance images in medical magnetic resonance imaging and magnetic resonance angiography (MRA). It is also used for making magnets, electronic gadgets, data storage disks, computer memory, color TV tubes, etc.

Gadolinium oxide was discovered by Jean Charles de Marignacn in 1880 by using spectroscopy. The name of the element was given in honor of scientist Johan Gadolin. The pure metal was first isolated by the French chemist Paul-Émile Lecoq de Boisbaudran in 1886.
Gadolinium Isotopes
Naturally occurring gadolinium contains six isotopes 154Gd, 155Gd, 156Gd, 157Gd, 158Gd, and 160Gd. It also contains one naturally occurring radioisotope, 152Gd. It also contains other 29 radioactive isotopes that can be prepared by various artificial nuclear reactions. The majority of these radioactive isotopes have half-lives shorter than 25 seconds.
The primary radioactive decay mode of gadolinium isotopes is electron capture or beta decay. Like other lanthanides, various radioactive isotopes of Gd are produced during nuclear fission reactions.
Properties
It is one of the light rare earth metals belonging to the lanthanide series in the periodic table. The metal Gd shows ferromagnetic properties and gadolinium is more ferromagnetic than iron at low temperatures. It also contains superconductive properties.
It crystallizes to form a hexagonal close-packed structure at room temperature. When heated above 1,235 °C, it transforms into a body-centered cubic crystal lattice.
| Gadolinium | |||
| Symbol | Gd | ||
| Discovery | Jean Charles Galissard de Marignac in 1880 | ||
| Name derived from | The honour of scientist Johan Gadolin | ||
| Common isotope | 64Gd158 | ||
| Oxidation states | +3 | ||
| CAS number | 7440-54-2 | ||
| Periodic properties | |||
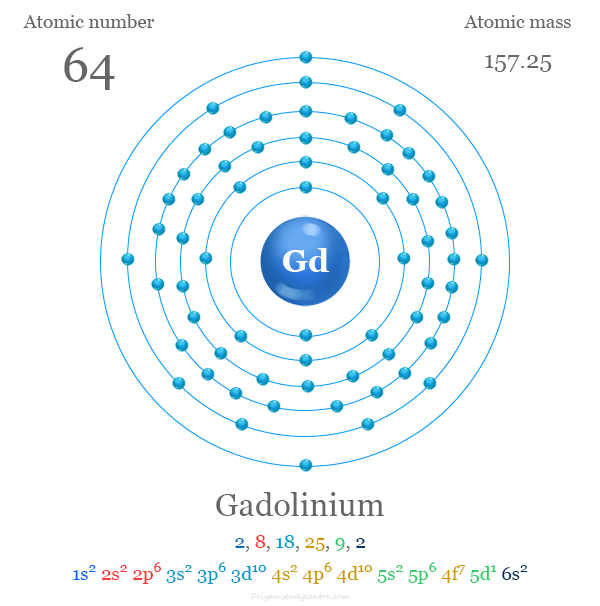
| Atomic number | 64 | ||
| Relative atomic mass | 157.25 | ||
| Electron per cell | 2, 8, 18, 25, 9, 2 | ||
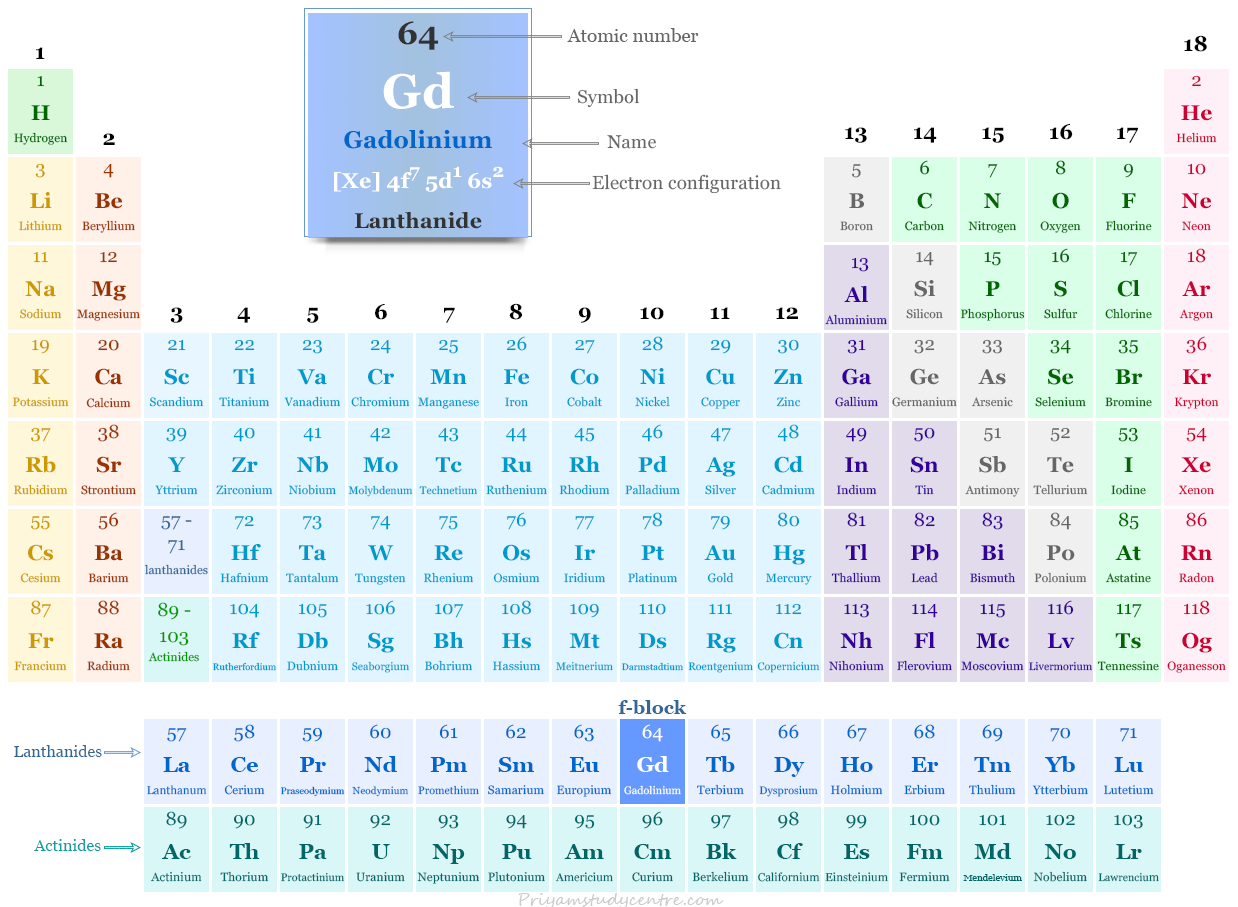
| Electronic Configuration | [Xe] 4f7 5d1 6s2 | ||
| Block | f-block | ||
| Group | Lanthanides | ||
| Period | 6 | ||
| Physical properties | |||
| State at 20 °C | Solid | ||
| Melting point | 1313 °C, 1586 K | ||
| Boiling point | 3273 °C, 3546 K | ||
| Molar heat capacity | 37.03 J mol−1 K−1 | ||
| Crystal structure | hexagonal close-packed (hcp) | ||
| Density | 7.90 g/cm3 | ||
| Heat of fusion | 10.05 kJ mol−1 | ||
| Heat of vaporization | 301.3 kJ mol−1 | ||
| Atomic properties | |||
| Atomic radius (non-bonded) | 2.34 Å | ||
| Covalent radius | 1.82 Å | ||
| Electronegativity | 1.20 (Pauling scale) | ||
| Electron affinity | Unknown | ||
| Ionization energy (kJ/mol) | 1st | 2nd | 3rd |
| 593.37 | 1166.51 | 1990.49 | |
Electron Configuration of Gadolinium
It contains 64 electrons that are arranged in the configuration of [Xe] 4f7 5d1 6s2. Gadolinium does not show a general trend of electronic configuration.
The 4f and 5d orbitals in Gd are about the same energy levels. Therefore, the Gd atom has a tendency to retain a half-filled 4f configuration. Generally, three valence electrons of gadolinium participate in chemical bonding due to the same reason.
Gadolinium in the Periodic Table
It is placed in the f-block of the periodic table. Gadolinium is a rare earth metal or lanthanide that lies between europium and terbium.
Chemical Properties
Like other lanthanides, it behaves as an active metal. It combines most of the periodic table elements to form Gd (III) derivatives.
Metallic gadolinium is relatively stable in dry air but it tarnishes quickly in humid air to form an oxide layer.
4 Gd + 3 O2 → 2 Gd2O3
The electropositive metal gadolinium dissolves slowly in cold water but quite quickly in warm water by the liberation of hydrogen and formation of Gd(OH)3.
2 Gd + 6 H2O → 2 Gd(OH)3 + 3 H2
It is a strong reducing agent that reduces various metal oxides to form their elemental state. It also combines with nitrogen, carbon, sulfur, phosphorus, boron, selenium, silicon, and arsenic at elevated temperatures to form their respective binary compounds.
The metal Gd is readily attacked by dilute sulfuric acid to form a colorless solution of Gd(III) ions.
2 Gd + 3 H2SO4 + 18 H2O → 2 [Gd(H2O)9]3+ + 3 SO4−2 + 3 H2
Facts About Gadolinium
- Gadolinium is a silvery, malleable, ductile metal that is used as a fluorescent.
- Like other rare earth elements, the elemental form of Gd is not found in nature. The primary source of this rare earth metal is gadolinite, monazite, and bastnasite.
- It is separated from other rare earth metals by solvent extraction process or ion exchange chromatography.
- In 1886, gadolinium was separated from its oxide by Lecoq de Boisbaudran. He named the rare earth metal in honor of Finnish Chemist Johan Gadolin.
- It is used in control rods of nuclear reactors to regulate nuclear fission reactions.
- It is injected into MRI patients to enhance image contrast.
- Free Gd+3 ion is considered to be a highly toxic species. Therefore, gadolinium MRI contrast agents are chelated compounds that are considered safe for human health.
Uses of Gadolinium
- Gadolinium is useful to improve the workability of iron and chromium alloys.
- It is alloyed with other metals for making magnets, electronic gadgets, data storage disks, and computer memory.
- It is a phosphor that is used in X-ray imaging systems.
- Gadolinium and its compounds are used for making green phosphors in color TV tubes.
- Solutions of organic gadolinium complexes are used for intravenous MRI contrast agents to enhance images in medical magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA) procedures
- Gadolinium compounds are very useful in magnetic resonance imaging (MRI) for diagnosing cancerous tumors.
- It is an excellent absorber for neutrons. Therefore, it may be used in control rods of nuclear power reactors.
- It is used for making gadolinium yttrium garnet which is used in many high-temperature devices such as microwaves and various optical components.
Toxicity
Free ion of gadolinium is considered to be highly toxic. The chelated compounds of Gd that we used in MRI contrast agents are considered to be safe for most persons. These chelating compounds of gadolinium may cause mild side effects like allergic reactions, nausea, and vomiting.