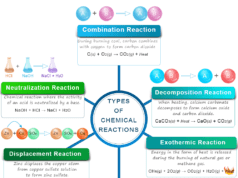
Chemical Equilibrium and Le Chatelier’s Principle
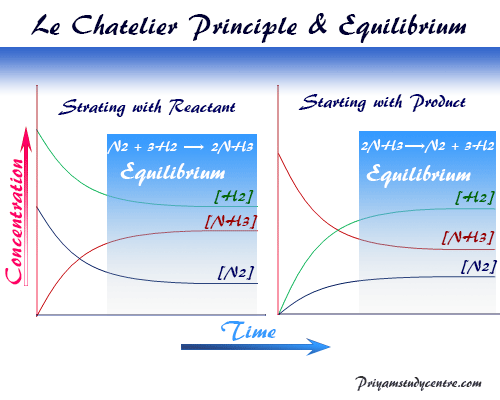
Le Chatelier’s principle in chemistry predicts the effect on the system at chemical equilibrium when some of the factors such as temperature, pressure, and concentration change. How the system behaves in chemistry or chemical science if any of these parameters of the system is altered was predicted by Le Chatelier in 1885 and Braun in 1886. Le Chatelier made the generalization that if a system is in equilibrium, a change in any of the factors that determine the condition of equilibrium will cause the equilibrium to shift in such a way as to minimize the effect of this change. Hence we use Le Chatelier’s principle which is sometimes called the principle of mobile equilibrium to express the thermodynamic effect of pressure, temperature, and concentration on chemical equilibrium for learning chemistry.
Application of Le Chatelier’s Principle
According to Le Chatelier’s principle, If the pressure increases in the system at chemical equilibrium, the volume would tend to diminish due to minimizing the effect of pressure.
In the synthesis of ammonia from hydrogen and nitrogen molecules, the volume would be decreased if the number of molecules decreases or more ammonia gas is formed.
When the pressure increases, the shift of the chemical reaction in such a direction where the sum of the stoichiometric coefficient of gas molecules is lowered.
In other words, an increase in pressure shifts the equilibrium from the low volume side to the high volume side.
Effect of Temperature on Equilibrium
Van’t Hoff equation numerically calculate the shift of equilibrium on temperature. However, Le Chatelier’s principle quantitative analysis of chemical equilibrium with the change of temperature.
According to Le Chatelier’s principle when the temperature increases, the chemical equilibrium will shift in the endothermic direction. Therefore, the equilibrium shifted from low enthalpy to high enthalpy side with the absorption or utilization of the specific heat.
The heat change or temperature change in chemical reactions also depends on the physical state like the gaseous, solid, or liquid state of the reacting component.
The oxidation of liquid benzene in the solution depends on the concentration of the solution. But for gaseous benzene in very dilute solutions heat of the reactions remains a constant value.
Application of Le Chatelier’s principle in Industrial Production of Ammonia
Let us illustrate this by elementary reaction for the formation of ammonia by the chemical bonding of nitrogen with the hydrogen atom.
For the synthesis of ammonia,
N2 + 3H2 ⇆ 2NH3 ΔH = −300 joule
where ΔH = ΣHproduct − ΣHreactant
In the above reaction, the enthalpy of the reactant side is greater than the product side.
- With the increases in temperature backward reaction favors where the equilibrium shifted to the higher entropy side. According to the Le Chatelier, with the increase in temperature backward reaction favors and the production of ammonia decreases.
- However, with the decrease in temperature, the equilibrium will shift in the exothermic direction. Since the shift will be high enthalpy side to the low enthalpy side. According to Le Chatelier’s principle with the decreases in temperature forward reaction favors and the production of ammonia increases.
Effect of Pressure on Equilibrium
According to Le Chatelier’s principle addition of inert gas or noble gas like helium, neon, argon, and krypton is done in two ways constant temperature and constant volume of the ideal system.
The addition of inert gas at constant volume can not affect the equilibrium. Since the concentration of the total reacting components remains unchanged according to Le Chatelier.
When inert gas is added to the system at constant pressure the volume of the reacting system increases. The total concentration or density decreased. Therefore, according to Le Chatelier’s principle, the system will move in the direction where the number of moles increases.
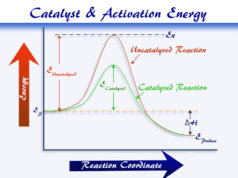
Effect of Catalyst on Equilibrium
A chemical catalyst is a foreign substance in chemical kinetics that enhances the speed of the chemical reaction itself remaining unaltered in mass and chemical composition. Catalysts can speed up the rate of reaction without affecting the equilibrium.
For example, oxidizing carbon monoxide to carbon dioxide in the presence of nitric oxide speeds up the rate. Hence Le Chatelier’s principle suggests that the reaction proceeds by lowering the activation energy to reach chemical equilibrium quickly.
Catalytic Poison Example
Sometimes a small quantity of foreign substances inhabits the reaction rate. These are called catalyst poison. In some cases, the poisons may form definite chemical compounds with catalyst atoms.
For example, a small quantity of arsenic destroys the catalytic activity of the platinum element by forming platinum arsenide in the sulfuric acid manufacturing process.
Question: What happens to the vapour pressure of a liquid, when a nonvolatile solute is dissolved in it according to the Le Chatelier?
Answer: The pure solution, the mole fraction x1 = 1. When the non-volatile solute is added to the solvent the mole fraction of the solvent changes from 1 to < 1.
To reduce these effects, according to Le Chatelier’s principle, the solvent is less vaporized. This leads to a lowering of the vapour pressure.
Le Chatelier’s Principle Facts
Although the equilibrium constant has no dimension and is independent of the pressure of reaction for the ideal gas or real gas molecules. However, Le Chatelier’s principle conclusions some special facts for reaction and product components.
For example, if the volume of the nonreactive system decreased by increasing a specific amount of the pressure. According to Le Chatelier’s principle, the chemical equilibrium shifted to the low-volume sides. Hence the pressure increases become less than that of the non-reactive system.
This shift in the equilibrium position of natural gases makes the reactive system have higher compressibility than the non-reactive one.
Similarly, if the fixed quantity of the heat supplied to the non-reacting system temperature of corresponding increases. The heat supplied does not increase the temperature so much. Since the equilibrium shifts to the higher enthalpy or free energy side.
This shift of equilibrium makes the heat much higher than in the non-reactive system. The reacting system is chosen as a heat storage medium according to Le Chatelier’s principle.