Calcium Element
Calcium is the chemical element or alkaline-earth metal of Group-2 (IIA) of the periodic table with the symbol Ca and atomic number 20. It is an essential element in the human body because Ca is used for building bones, clotting blood, contracting muscles, and beating our hearts. The electronic configuration of calcium suggests that the metal has two s-electrons available for chemical bonding and it shows a common oxidation number or state of +2. Due to two electrons in the valence s-orbital, calcium was placed in the s-block of the periodic table. About 90 percent of the calcium in the human body is used to make our bones and teeth. It is a silvery-white, lustrous, and relatively hard metal than alkali metals.
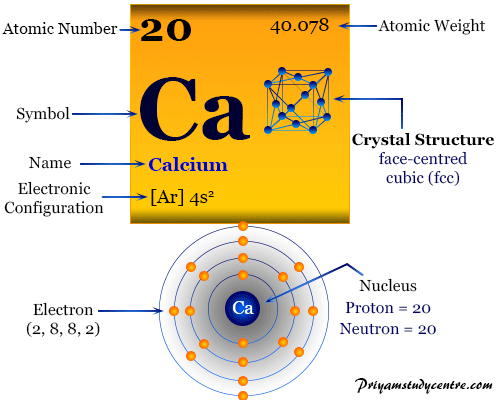
Properties of Calcium
It is formed a face-centered cubic crystal lattice with a higher melting point than alkali metals. It possesses a 4s2 valence shell electronic configuration over the previous noble gas argon. The s2 configuration leads to inclusion in Group-2 or IIA of the periodic table.
Some important properties of the metal are given below in the table,
| Properties of Calcium |
|||
| Atomic number | 20 | ||
| Electron per shell | 2, 8, 8, 2 | ||
| Atomic weight | 40.078 | ||
| Electronic Configuration | [Ar] 4s2 | ||
| Block | s-block | ||
| Period | 4 | ||
| Group | 2 | ||
| Melting point | 842 °C, 1548 °F | ||
| Boiling point | 1484 °C, 2703 °F | ||
| Molar heat capacity | 25.929 J mol−1 K−1 | ||
| Crystal structure | face-centered cubic (fcc) | ||
| Density | 1.55 g/cm3 | ||
| Common oxidation number or state | +2 | ||
| Electronegativity | 1.00 ( Pauling scale) | ||
| Ionization energy (kJ/mol) | 1st | 2nd | 3rd |
| 589.8 | 1145.4 | 4912.4 | |
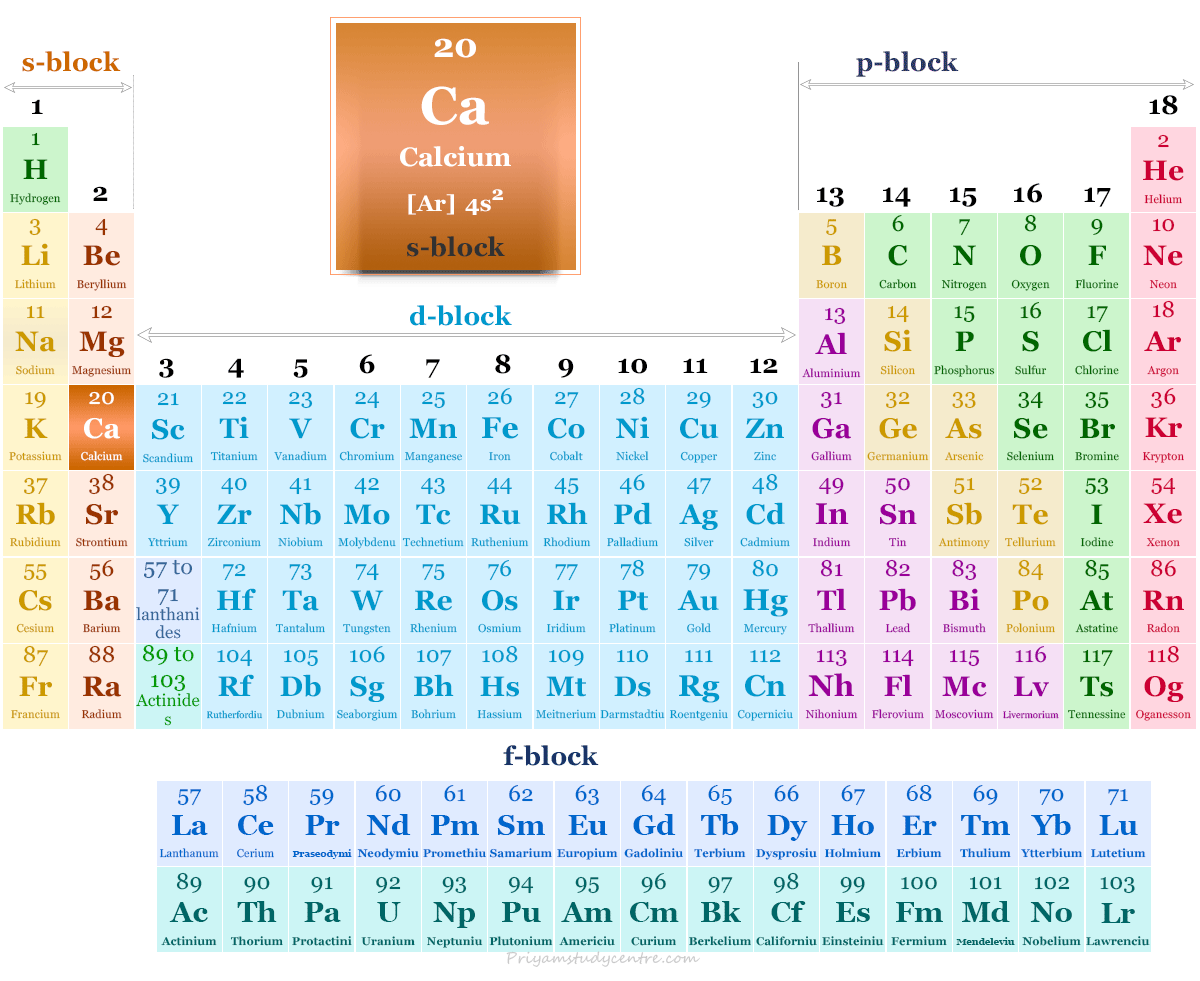
Calcium in the Periodic Table
Due to two electrons in the valence s-orbital, calcium was placed in the s-block of the periodic table with other group members beryllium, magnesium, strontium, barium, and radium.
Where is Calcium Found?
It is the fifth most abundant element (3.63 percent) in the earth’s crust. It is concentrated in minerals that are probably separated during the main stage of the crystallization of magma.
Weathering and leaching lead to the formation of minerals calcium carbonate (CaCO3) or limestone in earth soil. The very low solubility of CaCO3 is the most probable reason for the absence of a significant amount of Ca+2 ion in sea-water.
Gypsum (CaSO4, 2H2O), fluorspar (CaF2) and apatite [3Ca3(PO4)2, CaF2] are important minerals of calcium.
Sources of Calcium
Every day we lose calcium from our body through our skin, nails, hair, sweat, urine, and feces but our body cannot produce it. Therefore, we need to eat Ca-reached foods or vegetables that sustain the deficiency of Ca in our bodies.
Dairy products (milk, yogurt, cheese) and vegetables like green leaf, beans, broccoli, brussels sprouts, collards, kale, and mustard greens are the main sources of calcium for our daily requirements.
Isotopes of Ca
Naturally, occurring calcium is a mixture of five stable isotopes like 40Ca, 42Ca, 43Ca, 44Ca, and 46Ca. The radioactive isotope 48Ca (half-life of about 4.3 × 1019 years) can be considered stable.
It is the first lightest element that contains six naturally occurring isotopes.
Production Process
It is produced on a commercial scale by electrolysis of fused CaCl2. Some fluorspar (CaF2) was added due to the lower temperatures for fusion.
The electrolysis is carried out in a graphite pot serving as an electrode or anode. A water-cooled iron cathode hangs into the fused electrolyte.
The produced metal is deposited on the cathode in the form of a rod when the electrolysis proceeds. A layer of fused electrolyte protects the metal from oxidation.
Facts About Calcium
Grup-2 elements of the periodic table are characterized by two valence electrons in an s-shell of the atoms. These two electrons are generally involved together to show the uniform bivalency of the metal.
The properties of elements like Ca, St, Ba, Ra, and their compounds are systematically and gradually changed with the increasing atomic number.
Electropositive or metallic character and ionic radius increase from calcium to radium. Thermal stabilities of carbonates, nitrate, and sulfates are also increased from Ca to Ra.
Chemical Compounds
Calcium Hydride
Calcium hydride is formed by ionic bonding due to the high electropositive character and large size of the metal. CaH2 is a high melting crystalline solid, that conducts electricity in a fused state and liberates hydrogen on electrolysis.
The hydride is obtained during the direct combination of hydrogen with the metal. The reaction of CaH2 with water provides a ready source of hydrogen.
Calcium Oxide
All the oxides of Grup-2 elements are ionic with a high melting point but beryllium oxide is formed by covalent bonding.
The tendency to form peroxide and superoxide increases with the increasing atomic number of the Group-2 elements. However, calcium superoxide is formed by the reaction of metal peroxide with 30 percent hydrogen peroxide.
The oxides react with water to form hydroxides. The basic properties of Group-2 hydroxides increase from beryllium to barium.
Calcium Halide
All the crystalline halides of Ca are ionic in nature with a high melting point. CaF2 is a high melting (melting point 1418 °C) water-insoluble solid while chloride is a lower melting point and water-soluble.
The bromide and iodide have still lower meting and are higher soluble in water and alcohol.
What is Calcium Used For?
- Due to a strong chemical affinity for oxygen and sulfur, metallic calcium is widely used for making steel.
- It is an important reducing agent for the production of chromium, zirconium, thorium, and uranium.
- It is used as a scavenger in the steel industry to remove oxygen, sulfur, and phosphorus.
- Calcium is generally used in aluminum alloying to improve the strength of the metal.
- It is used to remove nitrogen from argon.
- It is also used for manufacturing the chemical compound, hydrolith (CaH2).
Function in the Human Body
About 90 percent of the metal in the human body is used to make our bones and teeth.
It not only plays a major structural role but also plays crucial regulatory functions like muscle construction, nerve transmission, and blood coagulation. The deficiency of calcium in the human body causes hypocalcemia, rickets, and osteoporosis.